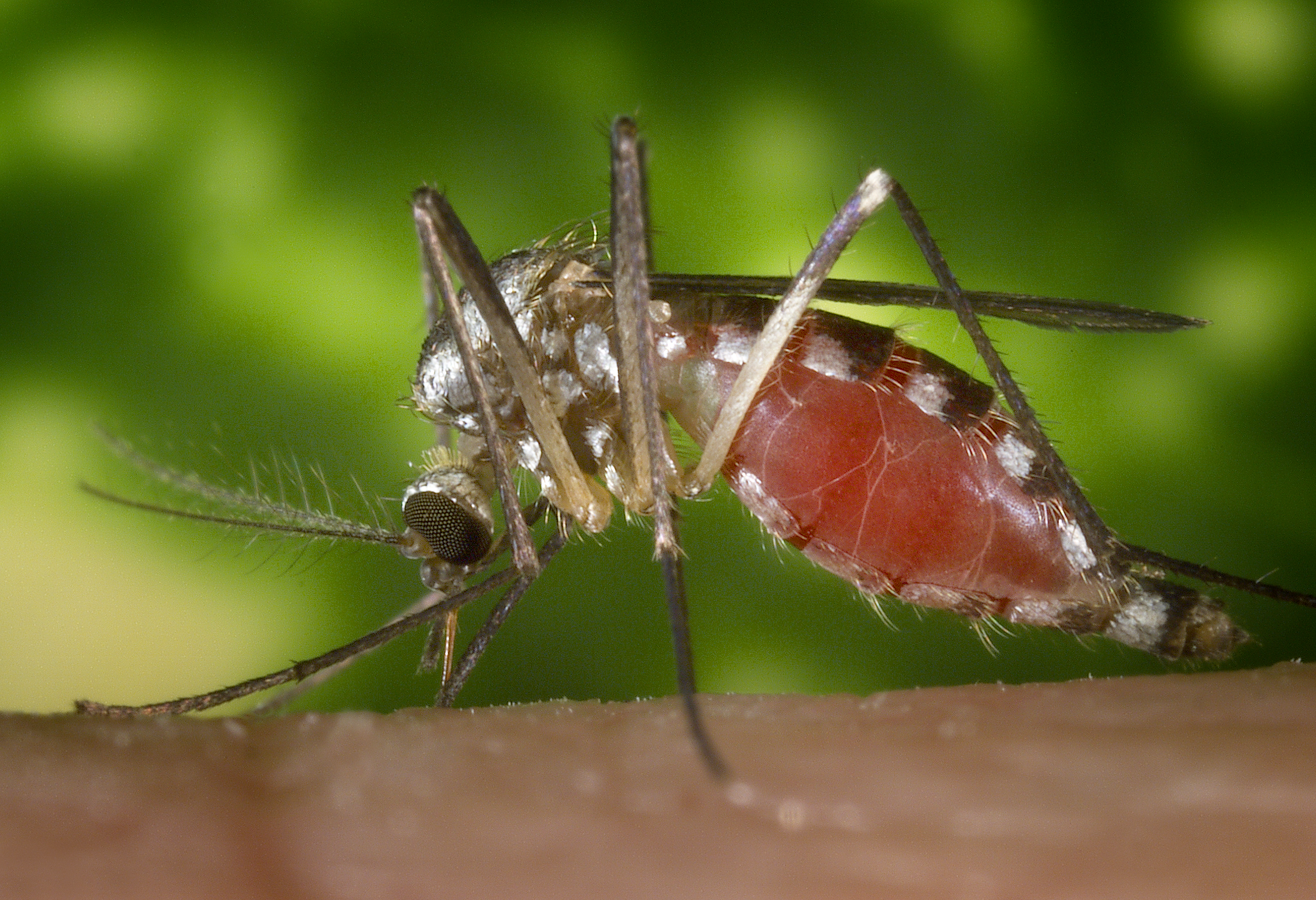
Encefalite é a inflamação do cérebro. A doença é de gravidade variável. Os sintomas mais comuns são dores de cabeça, febre, confusão, pescoço rígido e vómitos. Entre as possíveis complicações estão crises epilépticas, alucinações, dificuldade em falar, problemas de memória e problemas auditivos.
Introdução
O que você precisa saber de cara
Encefalite viral aguda causada pelo vírus La Crosse, transmitido por mosquitos. Afeta principalmente crianças, com sintomas como febre, dor de cabeça, convulsões e alterações neurológicas.
Escala de raridade
<1/50kMuito rara
1/20kRara
1/10kPouco freq.
1/5kIncomum
1/2k
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Nenhum gene associado encontrado
Os dados genéticos desta condição ainda estão sendo catalogados.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Encefalite da Califórnia
Selecione um estado ou use sua localização para ver resultados.
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Pesquisa e ensaios clínicos
Nenhum ensaio clínico registrado para esta condição.
Publicações mais relevantes
Replication competent La Crosse virus pseudotyped VSV vector system as vaccine and serological diagnostic reagent.
La Crosse encephalitis virus (LACV) is the second most frequently diagnosed arboviral cause of meningoencephalitis and the most common cause of pediatric viral encephalitis in the US. Multiple factors, including climate changes and invasive mosquito species adaptation contribute to the emerging spread of LACV in new areas. There is an expanding need for broader investigation on seroprevalence, development of novel antiviral drugs and vaccines for prevention of LACV infection. We pseudotyped replication-competent vesicular stomatitis virus (VSV) vectors with LACV surface glycoproteins using wild type (VSV-LACV) and attenuated (VSV-M51-LACV) backbones. Recombinant strains were tested as diagnostic tools in LACV serology and as potential live anti-LACV vaccine in vivo. The attenuated VSV-M51R-LACV vector was safe after direct intracranial inoculation in mice. Intraperitoneal immunization induced strong humoral response reaching average complete neutralization titer (VN100%) of 4640 in Balb/c and 2800 in interferon type I receptor knockout transgenic (Ifnarko-CD46Ge) mice respectively. Serum antibodies from vaccinated mice efficiently neutralized LACV with average titers between 229 and 2926 for the different LACV strains. We demonstrated that virus neutralization (VN) based on LACV-pseudotyped VSV is a valuable diagnostic assay for serodiagnostics of anti-LACV response in humans. VN titers in the range of 8-4096 were detected in sera from all individuals (n = 20) with confirmed infection. The assay also detected LACV-specific antibody response in cerebrospinal fluid of patients with CNS infection. VN was adapted for detection of neutralizing antibodies extracted from dried blood spots (DBS) samples with potential application in large scale LACV serologic surveillance. These data demonstrate that recombinant VSV-M51R-LACV is a valuable diagnostic tool as well as a promising live vaccine platform for immunoprophylaxis against LACV infection.
Neuropsychological and Functional Outcome after La Crosse Encephalitis in Marine Recruit: A Case Report and Literature Review.
La Crosse virus is a rare infection in humans that can cause encephalopathy and neuroinvasive disease following infection. While pediatric outcomes have been studied, long-term neuropsychological and functional outcomes in adults remain undocumented. This case report compares existing literature with the extended clinical course of a 19-year-old, young adult male Marine recruit who contracted LACV. The Marine presented with lethargy, altered mental status, and abnormal neuroimaging. After improvements in alertness and command following, he was transferred from acute care to an inpatient neurorehabilitation unit. Disability was measured with the Functional Independence Measure (FIM). Cognitive impairment was documented with neuropsychological assessments. After rehabilitation discharge, he was readmitted approximately 4.75 years postinfection for a respite stay and re-evaluated. FIM scores at admission to the neurorehabilitation facility reflected nearly total dependence. He emerged from a confusional state by 3.5 months postinfection but with severe and nearly global cognitive impairment. By 4.75 years, he showed improved independence in self-care and mobility. Neuropsychological gains were observed in the domains of memory, attention, and executive functioning. Persistent deficits in processing speed and motor dexterity remained. This case highlights the potential for significant long-term functional and cognitive recovery following severe La Crosse encephalitis with global impairment acutely. Periodic neuropsychological re-evaluation may be warranted after infection to inform updated rehabilitation goals and maximize functional independence.
A cap-dependent endonuclease inhibitor acts as a potent antiviral agent against La Crosse virus infection.
La Crosse virus (LACV) infection, the causative agent of La Crosse encephalitis, can lead to severe neurological symptoms and sequelae, particularly in children. Despite annual reports of neurologically symptomatic cases, no effective treatment has yet been established. Bunyaviruses, including LACV, utilize a cap-snatching mechanism for transcription, with a cap-dependent endonuclease (CEN) serving as a promising target for antiviral treatment. Specifically, we now demonstrate that a CEN inhibitor, carbamoyl pyridone carboxylic acid (CAPCA)-1, exhibits potent anti-LACV activity in vitro and in vivo. CAPCA-1 exhibited 50% effective concentration values below 1 µM in neuronal and non-neuronal cells, demonstrating a higher in vitro activity than the nucleoside analogs, ribavirin and favipiravir. Multiple passages of LACV in the presence of CAPCA-1 produced numerous amino acid mutations in the CEN active site. Notably, using a lethal infection model in mice, CAPCA-1 treatment reduced viral loads in the brain and extended the survival rate of LACV-infected mice. These findings highlight the potential of CEN inhibitors as treatment options for La Crosse encephalitis.
Development of FRET-based cap-snatching endonuclease assay.
The order Bunyavirales consists of over 300 species of segmented, negative-strand RNA viruses. These viruses have a tri-segmented genome (S, M, and L segments) that encodes the nucleocapsid protein (N protein), glycoprotein precursor, and RNA-dependent RNA polymerase (RdRp), respectively. The RdRp is a large protein (~420 kDa) responsible for synthesizing viral mRNA and replicating the viral genome. Bunyaviruses initiate transcription of viral mRNA through a unique cap-snatching mechanism. During this process, the N-terminal endonuclease domain of the RdRp cleaves host cell mRNA at the 5' terminus and uses the resulting capped mRNA fragment as a primer. This endonuclease domain exhibits a highly conserved structural architecture and is essential for establishing viral infection in host cells. Therefore, the N-terminal endonuclease domain represents a promising target for therapeutic intervention against Bunyaviruses, particularly at the early stages of the virus replication cycle. In this study, we developed a highly sensitive fluorescence resonance energy transfer-based in vitro assay to quantitatively examine the activity of the bacterially expressed and purified endonuclease domain of hantavirus RdRp. A 20-nucleotide synthetic RNA, labeled with a 6-FAM fluorophore at the 5' end and an Iowa Black quencher at the 3' end, generated a significant dequenched fluorescence signal upon cleavage by the purified endonuclease domain. Kinetic analysis revealed a half-life (t 1/2) of approximately 3 min for the reaction, achieving a signal-to-background ratio of approximately 31. Viruses belonging to the order Bunyavirales, including Hantaviruses, Crimean-Congo hemorrhagic fever virus, Rift Valley fever virus, Severe Fever with Thrombocytopenia Syndrome Virus, and La Crosse encephalitis virus, cause severe human illnesses with mortality rates in certain outbreaks reaching 50%, 10%-40%, 10%-20%, 6%-30%, and 1%, respectively. Currently, there are no Food and Drug Administration-approved vaccines or antiviral therapeutics available for these viruses. The highly efficient and cost-effective fluorescence resonance energy transfer-based in vitro endonuclease assay, having a quantitative fluorescence readout, can be optimized for high-throughput screening of chemical libraries to identify chemical inhibitors for the Bunyavirus cap-snatching endonuclease. The assay will be of critical importance for antiviral drug discovery against numerous negative-strand RNA viruses using cap-snatching mechanism for transcription initiation and replication of the RNA genome.
Current Status of La Crosse Virus in North America and Potential for Future Spread.
La Crosse virus (LACV) is an arthropod-borne RNA virus with substantial potential for future spread in North America. La Crosse virus is responsible for La Crosse encephalitis, a leading cause of arboviral encephalitis in children in the United States. Primarily transmitted by Aedes triseriatus (Eastern treehole) mosquitos and amplified by small mammal hosts, LACV has caused infections throughout the upper Midwest and, more recently, the mid-Atlantic and southeastern United States. Notably, in recent years, infections have also been identified increasingly in the Appalachian region. Anthropogenic and environmental factors have likely contributed to recent LACV spread, including the introduction of invasive vector species (especially Ae. albopictus), biotic interactions between and among vector and host species, land-use change, habitat disturbance, increased human travel and transport, and rising global temperatures. Prevention and control strategies, such as increased surveillance of vector and host populations, increased awareness among populations at risk for infection, and increased awareness among physicians are needed to limit future spread. Continued climate change with increases in global temperatures and erratic weather patterns may result in the expansion of competent mosquito vector species and thus could facilitate the geographic spread of LACV.
Publicações recentes
Replication competent La Crosse virus pseudotyped VSV vector system as vaccine and serological diagnostic reagent.
Neuropsychological and Functional Outcome after La Crosse Encephalitis in Marine Recruit: A Case Report and Literature Review.
A cap-dependent endonuclease inhibitor acts as a potent antiviral agent against La Crosse virus infection.
Development of FRET-based cap-snatching endonuclease assay.
Current Status of La Crosse Virus in North America and Potential for Future Spread.
📚 EuropePMC36 artigos no totalmostrando 20
Replication competent La Crosse virus pseudotyped VSV vector system as vaccine and serological diagnostic reagent.
Virus researchNeuropsychological and Functional Outcome after La Crosse Encephalitis in Marine Recruit: A Case Report and Literature Review.
Military medicineA cap-dependent endonuclease inhibitor acts as a potent antiviral agent against La Crosse virus infection.
Antimicrobial agents and chemotherapyDevelopment of FRET-based cap-snatching endonuclease assay.
Microbiology spectrumCurrent Status of La Crosse Virus in North America and Potential for Future Spread.
The American journal of tropical medicine and hygieneIdentification of Host Factors for Rift Valley Fever Phlebovirus.
VirusesPeridomestic conditions affect La Crosse virus entomological risk by modifying the habitat use patterns of its mosquito vectors.
Journal of vector ecology : journal of the Society for Vector EcologyDevelopment of a Community-Driven Mosquito Surveillance Program for Vectors of La Crosse Virus to Educate, Inform, and Empower a Community.
InsectsSpatial-temporal clusters of host-seeking Aedes albopictus, Aedes japonicus, and Aedes triseriatus collections in a La Crosse virus endemic county (Knox County, Tennessee, USA).
PloS oneHorizontal distribution affects the vertical distribution of native and invasive container-inhabiting Aedes mosquitoes within an urban landscape.
Journal of vector ecology : journal of the Society for Vector EcologyDouble Crossed: A Case of La Crosse Encephalitis.
Rhode Island medical journal (2013)Notes from the Field: Spatially Associated Coincident and Noncoincident Cases of La Crosse Encephalitis - North Carolina, 2002-2017.
MMWR. Morbidity and mortality weekly reportModeling a single season of Aedes albopictus populations based on host-seeking data in response to temperature and precipitation in eastern Tennessee.
Journal of vector ecology : journal of the Society for Vector EcologyLa Crosse Virus Vectors Are Host-Seeking and Ovipositing After 1700 H in Eastern Tennessee.
Journal of the American Mosquito Control AssociationEpidemiology of La Crosse Virus Emergence, Appalachia Region, United States.
Emerging infectious diseasesLa Crosse Encephalitis: A Persistent Arboviral Threat in North Carolina.
North Carolina medical journalLa Crosse Encephalitis: An Adult Case Series.
The American journal of medicineThe TAM receptor Mertk protects against neuroinvasive viral infection by maintaining blood-brain barrier integrity.
Nature medicineLa Crosse Encephalitis Virus Infection in Field-Collected Aedes albopictus, Aedes japonicus, and Aedes triseriatus in Tennessee.
Journal of the American Mosquito Control AssociationCemeteries are effective sites for monitoring la crosse virus (LACv) and these environments may play a role in LACv infection.
PloS oneAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Encefalite da Califórnia.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Encefalite da Califórnia
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Ainda não achamos doenças com sintomas parecidos o suficiente.
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Replication competent La Crosse virus pseudotyped VSV vector system as vaccine and serological diagnostic reagent.
- Neuropsychological and Functional Outcome after La Crosse Encephalitis in Marine Recruit: A Case Report and Literature Review.
- A cap-dependent endonuclease inhibitor acts as a potent antiviral agent against La Crosse virus infection.
- Development of FRET-based cap-snatching endonuclease assay.
- Current Status of La Crosse Virus in North America and Potential for Future Spread.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:83483(Orphanet)
- MONDO:0019378(MONDO)
- GARD:10925(GARD (NIH))
- Busca completa no PubMed(PubMed)
- Q2713959(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar