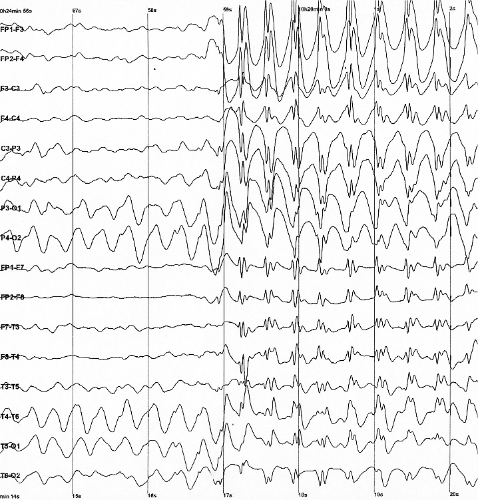
Uma síndrome rara de epilepsia genética. Ela se manifesta por convulsões sem febre em bebês recém-nascidos que, fora isso, são saudáveis. Essas convulsões começam nos primeiros dias de vida.
Introdução
O que você precisa saber de cara
Uma síndrome rara de epilepsia genética. Ela se manifesta por convulsões sem febre em bebês recém-nascidos que, fora isso, são saudáveis. Essas convulsões começam nos primeiros dias de vida.
Escala de raridade
<1/50kMuito rara
1/20kRara
1/10kPouco freq.
1/5kIncomum
1/2k
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Partes do corpo afetadas
+ 6 sintomas em outras categorias
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 23 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Genes associados
2 genes identificados com associação a esta condição. Padrão de herança: Autosomal dominant.
Pore-forming subunit of the voltage-gated potassium (Kv) M-channel which is responsible for the M-current, a key controller of neuronal excitability (PubMed:24277843, PubMed:28793216, PubMed:9836639). M-channel is composed of pore-forming subunits KCNQ2 and KCNQ3 assembled as heterotetramers (PubMed:10781098, PubMed:14534157, PubMed:32884139, PubMed:37857637, PubMed:9836639). The native M-current has a slowly activating and deactivating potassium conductance which plays a critical role in determ
Cell membrane
Seizures, benign familial neonatal 1
A disorder characterized by clusters of seizures occurring in the first days of life. Most patients have spontaneous remission by 12 months of age and show normal psychomotor development. Some rare cases manifest an atypical severe phenotype associated with epileptic encephalopathy and psychomotor retardation. The disorder is distinguished from benign familial infantile seizures by an earlier age at onset. In some patients, neonatal convulsions are followed later in life by myokymia, a benign condition characterized by spontaneous involuntary contractions of skeletal muscles fiber groups that can be observed as vermiform movement of the overlying skin. Electromyography typically shows continuous motor unit activity with spontaneous oligo- and multiplet-discharges of high intraburst frequency (myokymic discharges). Some patients may have isolated myokymia.
Pore-forming subunit of the voltage-gated potassium (Kv) M-channel which is responsible for the M-current, a key controller of neuronal excitability (PubMed:16319223, PubMed:27564677, PubMed:28793216, PubMed:9872318). M-channel is composed of pore-forming subunits KCNQ2 and KCNQ3 assembled as heterotetramers (PubMed:14534157, PubMed:16319223, PubMed:27564677, PubMed:9872318). The native M-current has a slowly activating and deactivating potassium conductance which plays a critical role in determ
Cell membrane
Seizures, benign familial neonatal 2
A disorder characterized by clusters of seizures occurring in the first days of life. Most patients have spontaneous remission by 12 months of age and show normal psychomotor development. The disorder is distinguished from benign familial infantile seizures by an earlier age at onset.
Variantes genéticas (ClinVar)
1,487 variantes patogênicas registradas no ClinVar.
Vias biológicas (Reactome)
2 vias biológicas associadas aos genes desta condição.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Epilepsia neonatal auto-limitada
Selecione um estado ou use sua localização para ver resultados.
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Pesquisa e ensaios clínicos
Nenhum ensaio clínico registrado para esta condição.
Publicações mais relevantes
The Clinical and Genetic Landscape of a French Multicenter Cohort of 2563 Epilepsy Patients Referred for Genetic Diagnosis.
Epileptic disorders are a heterogeneous group of neurological conditions, with many cases linked to monogenic causes, particularly in developmental and epileptic encephalopathies (DEE). Identifying pathogenic variants aids treatment, prognosis, and family planning. In France, genetic testing is coordinated through the EpiGene network. We analyzed clinical and genetic data from 2563 epilepsy patients referred to four diagnostic labs (2016-2023). Epilepsy syndromes were classified via pre-test questionnaires, and genotyping used various gene panels, including a 68-gene core panel. Multivariate logistic regression assessed diagnostic rates and genotype-phenotype correlations. Overall, 27.0% of patients had pathogenic/likely pathogenic variants, mainly within the core panel (24%). SCN1A and KCNQ2 were the most frequently mutated genes. Diagnostic yield varied by syndrome, with Dravet Syndrome Spectrum (DSS) and early-infantile DEE (EIDEE) showing the highest rates (41% and 34%, respectively). Genetic heterogeneity differed across syndromes, from DSS (predominantly SCN1A) to Infantile Epileptic Spasms Syndrome (IESS, 12%), involving ≥ 26 genes. Outside DEE, self-limited neonatal epilepsy (SeLNE) had the highest yield (50%). Earlier seizure onset was associated with a higher likelihood of a positive molecular diagnosis, whereas intellectual disability severity and drug resistance were not independently predictive of diagnostic outcome. Genotype-phenotype correlations highlighted that objective clinical data (e.g., age of onset) can outperform syndrome labels (e.g., EIDEE) in predicting diagnosis. This large cohort study refines the genetic landscape of epilepsy, informs classification challenges, and enhances genetic testing strategies, ultimately improving patient care and future research directions.
Novel KCNQ2 Variants Related to a Variable Phenotypic Spectrum Ranging from Epilepsy with Auditory Features to Severe Developmental and Epileptic Encephalopathies.
Pathogenic KCNQ2 variants are associated with neonatal epilepsies, ranging from self-limited neonatal epilepsy to KCNQ2-developmental and epileptic encephalopathy (DEE). In this study, next-generation sequencing was performed, applying a panel of 142 epilepsy genes on three unrelated individuals and affected family members, showing a wide variability in the epileptic spectrum. The genetic analysis revealed two likely pathogenic missense variants (c.1378G>A and c.2251T>G) and the already-reported pathogenic splice site (c.1631+1G>A) in KCNQ2 (HGNC:6296). The phenotypes observed in the affected members of family 1, which shared the c.2251T>G variant, were epilepsy with auditory features (EAFs), focal epilepsy, and generalized epilepsy, and none of them suffered from neonatal seizures. The gene panel contained further genes related to EAFs (LGI1, RELN, SCN1A, and DEPDC5), which were tested with negative results. The phenotypes observed in family 2 members, sharing the splice site variant, were neonatal seizures and focal epilepsy in childhood. The last unrelated proband, harboring the de novo missense c.1378G>A, presented a clinical phenotype consistent with DEE. In conclusion, we identified two unreported KCNQ2 variants, and report a proband with EAFs and individuals without typical KCNQ2 neonatal seizures. Our study underscores the extreme variability in the phenotypic spectrum of KCNQ2-related epilepsies and unveils the prospect of its inclusion in screening panels for EAFs.
Clinical and genetic analysis of 18 patients with KCNQ2 mutations from South China.
We aimed to delineate the genotype and phenotype of patients with KCNQ2 mutations from South China. Clinical manifestations and characteristics of KCNQ2 mutations of patients from South China were analyzed. Previous patients with mutations detected in this study were reviewed. Eighteen epilepsy patients with KCNQ2 mutations, including seven self-limited neonatal epilepsy (SeLNE), two self-limited infantile epilepsy (SeLIE) and nine developmental and epileptic encephalopathy (DEE) were enrolled. The age of onset (p=0.006), mutation types (p=0.029), hypertonia (p=0.000), and seizure offset (p=0.029) were different in self-limited epilepsy (SeLE) and DEE. De novo mutations were mainly detected in DEE patients (p=0.026). The mutation position, EEG or the age of onset were not predictive for the seizure or ID/DD outcome in DEE, while the development of patients free of seizures was better than that of patients with seizures (p=0.008). Sodium channel blockers were the most effective anti-seizure medication, while the age of starting sodium channel blockers did not affect the seizure or development offset. We first discovered the seizure recurrence ratio in SeLNE/SeLIE was 23.1% in South China. Four novel mutations (c.790T>C, c.355_363delGAGAAGAG, c.296+2T>G, 20q13.33del) were discovered. Each of eight mutations (c.1918delC, c.1678C>T, c.683A>G, c.833T>C, c.868G>A, c.638G>A, c.997C>T, c.830C>T) only resulted in SeLE or DEE, while heterogeneity was also found. Six patients in this study have enriched the known phenotype caused by the mutations (c.365C>T, c.1A>G, c.683A>G, c.833T>C, c.830C>T, c.1678C>T). This research has expanded known phenotype and genotype of KCNQ2-related epilepsy, and the different clinical features of SeLE and DEE from South China.
Neurodevelopmental outcome of neonatal seizures: A longitudinal study.
Neonatal seizures (NS) are the most common neurological emergency in the neonatal period. The International League Against Epilepsy (ILAE) proposed a new classification of NS based on semiology and highlighted the correlation between semiology and aetiology. However, neurodevelopmental outcomes have not been comprehensively evaluated based on this new classification. To evaluate neurodevelopmental outcomes and potential risk factors for severe outcomes in NS. Patients with video electroencephalogram confirmed NS were evaluated. Seizure aetiology, cerebral magnetic resonance imaging (MRI) data, background electroencephalograms data, general movements, and neurodevelopmental outcomes were analysed. Severe outcomes were one of the following: death, cerebral palsy, Griffiths developmental quotient <70, epilepsy, deafness, or blindness. A total of 74 neonates were evaluated: 62 (83.8 %) with acute provoked NS (primarily hypoxic-ischaemic encephalopathy), and 12 (16.2 %) with neonatal-onset epilepsies (self-limited neonatal epilepsy, developmental and epileptic encephalopathy, cerebral malformations). Of these, 32 (43.2 %) had electrographic seizures, while 42 (56.7 %) had electroclinical seizures - 38 (90.5 %) were motor (42.1 % clonic) and 4 (9.5 %) were non-motor phenomena. Severe outcomes occurred in 33 of the 74 (44.6 %) participants. In multivariate analysis, neonatal-onset epilepsies (odds ratio [OR]: 1.3; 95 % confidence interval [CI]: 1.1-1.6), status epilepticus (OR: 5.4; 95 % CI: 1.5-19.9), and abnormal general movements (OR: 3.4; 95 % CI: 1.9-7.6) were associated with severe outcomes. At present, hypoxic-ischaemic encephalopathy remains the most frequent aetiology of NS. The prognosis of neonatal-onset epilepsies was worse than that of acute provoked NS, and status epilepticus was the most predictive factor for adverse outcomes.
Retrospective study on neonatal seizures in a tertiary center of northern Italy after ILAE classification: Incidence, seizure type, EEG and etiology.
We aimed to evaluate epidemiology, seizure type, EEG, and etiology of neonatal seizures (NS) in a tertiary neonatal intensive care unit. Data on infants with a neurophysiological confirmation of NS were collected between 2009 and 2022. Seizure types and epileptic syndromes were classified by the ILAE classification and EEG by the Italian Neonatal Seizure Collaborative Network (INNESCO) score. Out of 91,253 neonates, 145 presented with NS; 69.7 % were born at term and 30.3 % were preterm infants. The incidence of NS in neonates born at our center was 1.2 per 1,000 live newborns (96/80697 neonates) while in the entire neonatal population admitted to our center it was 1.6 per 1,000 live births, increasing with lower preterm age. Compared to previous studies, we found a lower proportion of hypoxic-ischemic encephalopathy (HIE) (23.4 %) and a higher rate of genetic contribution (26.2 %). The infection rate was higher in preterm (31.8 %) than in full term (9.9 %) infants. Electrographic seizures were associated with acute provoked seizures (35.9 %), preterm age (52.3 %), and HIE (52.9 %). Vascular etiology was associated with focal clonic seizures (56.8 %). Non-structural neonatal genetic epilepsy was associated with sequential seizures (68.2 %), particularly KCNQ2 and SCN2A epilepsy. Background EEG was abnormal in all HIE, infections (85.7 %) and metabolic NS (83.3 %). In genetic epilepsy, background EEG depended on the epileptic syndrome: normal in 80 % of self-limited neonatal epilepsy and abnormal in 77.8 % of developmental and epileptic encephalopathy. Electroclinical seizures were associated with focal onset, while electrographic seizures correlated with a multifocal onset. A low incidence of HIE and a high incidence of genetic etiology were observed in our cohort of NS. Seizure type and EEG features are fundamental to address etiology.
Publicações recentes
The Clinical and Genetic Landscape of a French Multicenter Cohort of 2563 Epilepsy Patients Referred for Genetic Diagnosis.
Novel KCNQ2 Variants Related to a Variable Phenotypic Spectrum Ranging from Epilepsy with Auditory Features to Severe Developmental and Epileptic Encephalopathies.
Retrospective study on neonatal seizures in a tertiary center of northern Italy after ILAE classification: Incidence, seizure type, EEG and etiology.
Clinical and genetic analysis of 18 patients with KCNQ2 mutations from South China.
Neurodevelopmental outcome of neonatal seizures: A longitudinal study.
📚 EuropePMCmostrando 9
The Clinical and Genetic Landscape of a French Multicenter Cohort of 2563 Epilepsy Patients Referred for Genetic Diagnosis.
European journal of neurologyNovel KCNQ2 Variants Related to a Variable Phenotypic Spectrum Ranging from Epilepsy with Auditory Features to Severe Developmental and Epileptic Encephalopathies.
International journal of molecular sciencesRetrospective study on neonatal seizures in a tertiary center of northern Italy after ILAE classification: Incidence, seizure type, EEG and etiology.
Epilepsy & behavior : E&BClinical and genetic analysis of 18 patients with KCNQ2 mutations from South China.
The Turkish journal of pediatricsNeurodevelopmental outcome of neonatal seizures: A longitudinal study.
European journal of paediatric neurology : EJPN : official journal of the European Paediatric Neurology SocietyClinical analysis and functional characterization of KCNQ2-related developmental and epileptic encephalopathy.
Frontiers in molecular neuroscienceCase Report: Effect of Targeted Therapy With Carbamazepine in KCNQ2 Neonatal Epilepsy.
Frontiers in neurologyIn vitro and in vivo anti-epileptic efficacy of eslicarbazepine acetate in a mouse model of KCNQ2-related self-limited epilepsy.
British journal of pharmacologyAutism and developmental disability caused by KCNQ3 gain-of-function variants.
Annals of neurologyAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Epilepsia neonatal auto-limitada.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Epilepsia neonatal auto-limitada
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- The Clinical and Genetic Landscape of a French Multicenter Cohort of 2563 Epilepsy Patients Referred for Genetic Diagnosis.
- Novel KCNQ2 Variants Related to a Variable Phenotypic Spectrum Ranging from Epilepsy with Auditory Features to Severe Developmental and Epileptic Encephalopathies.
- Clinical and genetic analysis of 18 patients with KCNQ2 mutations from South China.
- Neurodevelopmental outcome of neonatal seizures: A longitudinal study.European journal of paediatric neurology : EJPN : official journal of the European Paediatric Neurology Society· 2024· PMID 38324990mais citado
- Retrospective study on neonatal seizures in a tertiary center of northern Italy after ILAE classification: Incidence, seizure type, EEG and etiology.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:1949(Orphanet)
- MONDO:0016027(MONDO)
- Epilepsia(PCDT · Ministério da Saúde)
- GARD:1519(GARD (NIH))
- Variantes catalogadas(ClinVar)
- Busca completa no PubMed(PubMed)
- Q4887956(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar