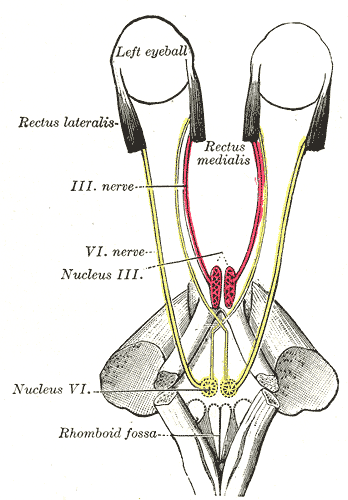
A paralisia facial congênita hereditária isolada (IHCFP) é uma doença neurológica extremamente rara, que se acredita ser causada por um desenvolvimento anormal do núcleo facial (uma parte do cérebro) e/ou do nervo facial. Até o momento, foi relatada em menos de 10 famílias. Ela se manifesta como uma paralisia facial que não piora com o tempo, afeta apenas o rosto e pode atingir um lado ou os dois lados, de forma igual ou desigual. As diferentes ramificações do nervo facial podem ser atingidas de forma desigual.
Introdução
O que você precisa saber de cara
A paralisia facial congênita hereditária isolada (IHCFP) é uma doença neurológica extremamente rara, que se acredita ser causada por um desenvolvimento anormal do núcleo facial (uma parte do cérebro) e/ou do nervo facial. Até o momento, foi relatada em menos de 10 famílias. Ela se manifesta como uma paralisia facial que não piora com o tempo, afeta apenas o rosto e pode atingir um lado ou os dois lados, de forma igual ou desigual. As diferentes ramificações do nervo facial podem ser atingidas de forma desigual.
Escala de raridade
<1/50kMuito rara
1/20kRara
1/10kPouco freq.
1/5kIncomum
1/2k
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Partes do corpo afetadas
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 3 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Nenhum gene associado encontrado
Os dados genéticos desta condição ainda estão sendo catalogados.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Paralisia facial isolada hereditária congênita
Centros de Referência SUS
24 centros habilitados pelo SUS para Paralisia facial isolada hereditária congênita
Centros para Paralisia facial isolada hereditária congênita
Detalhes dos centros
Hospital Universitário Prof. Edgard Santos (HUPES)
R. Dr. Augusto Viana, s/n - Canela, Salvador - BA, 40110-060 · CNES 0003808
Serviço de Referência
Hospital Infantil Albert Sabin
R. Tertuliano Sales, 544 - Vila União, Fortaleza - CE, 60410-794 · CNES 2407876
Serviço de Referência
Hospital de Apoio de Brasília (HAB)
AENW 3 Lote A Setor Noroeste - Plano Piloto, Brasília - DF, 70684-831 · CNES 0010456
Serviço de Referência
Hospital Estadual Infantil e Maternidade Alzir Bernardino Alves (HIABA)
Av. Min. Salgado Filho, 918 - Soteco, Vila Velha - ES, 29106-010 · CNES 6631207
Serviço de Referência
Hospital das Clínicas da UFG
Rua 235 QD. 68 Lote Área, Nº 285, s/nº - Setor Leste Universitário, Goiânia - GO, 74605-050 · CNES 2338424
Serviço de Referência
Hospital Universitário da UFJF
R. Catulo Breviglieri, Bairro - s/n - Santa Catarina, Juiz de Fora - MG, 36036-110 · CNES 2297442
Atenção Especializada
Hospital das Clínicas da UFMG
Av. Prof. Alfredo Balena, 110 - Santa Efigênia, Belo Horizonte - MG, 30130-100 · CNES 2280167
Serviço de Referência
Hospital Universitário Julio Müller (HUJM)
R. Luis Philippe Pereira Leite, s/n - Alvorada, Cuiabá - MT, 78048-902 · CNES 2726092
Atenção Especializada
Hospital Universitário João de Barros Barreto
R. dos Mundurucus, 4487 - Guamá, Belém - PA, 66073-000 · CNES 2337878
Serviço de Referência
Hospital Universitário Lauro Wanderley (HULW)
R. Tabeliao Estanislau Eloy, 585 - Castelo Branco, João Pessoa - PB, 58050-585 · CNES 0002470
Atenção Especializada
Instituto de Medicina Integral Prof. Fernando Figueira (IMIP)
R. dos Coelhos, 300 - Boa Vista, Recife - PE, 50070-902 · CNES 0000647
Serviço de Referência
Hospital Pequeno Príncipe
R. Des. Motta, 1070 - Água Verde, Curitiba - PR, 80250-060 · CNES 3143805
Serviço de Referência
Hospital Universitário Regional de Maringá (HUM)
Av. Mandacaru, 1590 - Parque das Laranjeiras, Maringá - PR, 87083-240 · CNES 2216108
Atenção Especializada
Hospital de Clínicas da UFPR
R. Gen. Carneiro, 181 - Alto da Glória, Curitiba - PR, 80060-900 · CNES 2364980
Serviço de Referência
Hospital Universitário Pedro Ernesto (HUPE-UERJ)
Blvd. 28 de Setembro, 77 - Vila Isabel, Rio de Janeiro - RJ, 20551-030 · CNES 2280221
Serviço de Referência
Instituto Nacional de Saúde da Mulher, da Criança e do Adolescente Fernandes Figueira (IFF/Fiocruz)
Av. Rui Barbosa, 716 - Flamengo, Rio de Janeiro - RJ, 22250-020 · CNES 2269988
Serviço de Referência
Hospital São Lucas da PUCRS
Av. Ipiranga, 6690 - Jardim Botânico, Porto Alegre - RS, 90610-000 · CNES 2232928
Serviço de Referência
Hospital de Clínicas de Porto Alegre (HCPA)
Rua Ramiro Barcelos, 2350 Bloco A - Av. Protásio Alves, 211 - Bloco B e C - Santa Cecília, Porto Alegre - RS, 90035-903 · CNES 2237601
Serviço de Referência
Hospital Universitário da UFSC (HU-UFSC)
R. Profa. Maria Flora Pausewang - Trindade, Florianópolis - SC, 88036-800 · CNES 2560356
Serviço de Referência
Hospital das Clínicas da FMUSP
R. Dr. Ovídio Pires de Campos, 225 - Cerqueira César, São Paulo - SP, 05403-010 · CNES 2077485
Serviço de Referência
Hospital de Base de São José do Rio Preto
Av. Brg. Faria Lima, 5544 - Vila Sao Jose, São José do Rio Preto - SP, 15090-000 · CNES 2079798
Atenção Especializada
Hospital de Clínicas da UNICAMP
R. Vital Brasil, 251 - Cidade Universitária, Campinas - SP, 13083-888 · CNES 2748223
Serviço de Referência
Hospital de Clínicas de Ribeirão Preto (HCRP-USP)
R. Ten. Catão Roxo, 3900 - Vila Monte Alegre, Ribeirão Preto - SP, 14015-010 · CNES 2082187
Serviço de Referência
UNIFESP / Hospital São Paulo
R. Napoleão de Barros, 715 - Vila Clementino, São Paulo - SP, 04024-002 · CNES 2688689
Serviço de Referência
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Pesquisa e ensaios clínicos
Nenhum ensaio clínico registrado para esta condição.
Publicações mais relevantes
Magnetic resonance imaging of developmental facial paresis: a spectrum of complex anomalies.
Despite its clinical implications, the MRI features of developmental facial paresis (DFP) were described in a few case reports. This study aims to describe MRI features of DFP in relation to the embryological development with a proposed radiological new grading system. The clinical records and MRI of the brain and internal auditory canal of 11 children with DFP were retrospectively reviewed. The following sequences were analyzed: axial, oblique sagittal SPACE of the internal auditory canal and brainstem; axial T2, T1WI and coronal T2WI of the brain. The severity of the maldevelopment of the seventh nerve was graded from 0 to 4: 0 = no abnormalities, 1 = unilateral facial nerve hypoplasia, 2 = unilateral facial nerve aplasia, 3 = aplasia or hypoplasia involving facial nerves on both sides, and 4 = facial nerve aplasia or hypoplasia associated with other cranial nerve palsy. Isolated facial nerve palsy was diagnosed in seven patients. It was of grade 1 in five and grade 3 in two. Hypoplasia of the nerve with interrupted course was encountered in two cases. Other associated cranial nerve abnormalities (grade 4) were seen in four patients; two of them were diagnosed previously as Moebius syndrome. In addition to inner ear anomalies, middle and external ear and parotid gland anomalies were described. To our knowledge, this is the largest series of patients with DFP that represents a continuum of isolated and combined malformations. Understanding of embryological basis can give insights into the anomalous development of the facial nerve.
A novel homozygous HOXB1 mutation in a Turkish family with hereditary congenital facial paresis.
Hereditary congenital facial paresis (HCFP) is characterized by isolated dysfunction of the facial nerve (CN VII) due to congenital cranial dysinnervation disorders. HCFP has genetic heterogeneity and HOXB1 is the first identified gene. We report the clinical, radiologic and molecular investigations of three patients admitted for HCFP in a large consanguineous Turkish family. High-throughput sequencing and Sanger sequencing of all patients revealed a novel homozygous mutation p.Arg230Trp (c.688C>T) within the HOXB1 gene. The report of the mutation brings the total number of HOXB1 mutations identified in HCFP to four. The results of this study emphasize that in individuals with congenital facial palsy accompanied by hearing loss and dysmorphic facial features, HOXB1 mutation causing HCFP should be kept in mind.
An exome sequencing study of Moebius syndrome including atypical cases reveals an individual with CFEOM3A and a TUBB3 mutation.
Moebius syndrome is characterized by congenital unilateral or bilateral facial and abducens nerve palsies (sixth and seventh cranial nerves) causing facial weakness, feeding difficulties, and restricted ocular movements. Abnormalities of the chest wall such as Poland anomaly and variable limb defects are frequently associated with this syndrome. Most cases are isolated; however, rare families with autosomal dominant transmission with incomplete penetrance and variable expressivity have been described. The genetic basis of this condition remains unknown. In a cohort study of nine individuals suspected to have Moebius syndrome (six typical, three atypical), we performed whole-exome sequencing to try to identify a commonly mutated gene. Although no such gene was identified and we did not find mutations in PLXND1 and REV3L, we found a de novo heterozygous mutation, p.E410K, in the gene encoding tubulin beta 3 class III (TUBB3), in an individual with atypical Moebius syndrome. This individual was diagnosed with near-complete ophthalmoplegia, agenesis of the corpus callosum, and absence of the septum pellucidum. No substantial limb abnormalities were noted. Mutations in TUBB3 have been associated with complex cortical dysplasia and other brain malformations and congenital fibrosis of extraocular muscles type 3A (CFEOM3A). Our report highlights the overlap of genetic etiology and clinical differences between CFEOM and Moebius syndrome and describes our approach to identifying candidate genes for typical and atypical Moebius syndrome.
Homozygous HOXB1 loss-of-function mutation in a large family with hereditary congenital facial paresis.
Hereditary congenital facial paresis (HCFP) belongs to the congenital cranial dysinnervation disorders. HCFP is characterized by the isolated dysfunction of the seventh cranial nerve and can be associated with hearing loss, strabismus, and orofacial anomalies. Möbius syndrome shares facial palsy with HCFP, but is additionally characterized by limited abduction of the eye(s). Genetic heterogeneity has been documented for HCFP as one locus mapped to chromosome 3q21-q22 (HCFP1) and a second to 10q21.3-q22.1 (HCFP2). The only known causative gene for HCFP is HOXB1 (17q21; HCFP3), encoding a homeodomain-containing transcription factor of the HOX gene family, which are master regulators of early developmental processes. The previously reported HOXB1 mutations change arginine 207 to another residue in the homeodomain and alter binding capacity of HOXB1 for transcriptional co-regulators and DNA. We performed whole exome sequencing in HCFP-affected individuals of a large consanguineous Moroccan family. The homozygous nonsense variant c.66C>G/p.(Tyr22*) in HOXB1 was identified in the four patients with HCFP and ear malformations, while healthy family members carried the mutation in the heterozygous state. This is the first disease-associated HOXB1 mutation with a likely loss-of-function effect suggesting that all HOXB1 variants reported so far also have severe impact on activity of this transcriptional regulator. © 2016 Wiley Periodicals, Inc.
A new hereditary congenital facial palsy case supports arg5 in HOX-DNA binding domain as possible hot spot for mutations.
Moebius syndrome (MBS) is a rare congenital disorder characterized by rhombencephalic mal development, mainly presenting with facial palsy with limited gaze abduction. Most cases are sporadic, possibly caused by a combination of environmental and genetic factors; however, no proven specific associations have been yet established. Hereditary congenital facial palsy (HCFP) is an autosomal dominant congenital dysinnervation syndrome, recognizable by the isolated dysfunction of the seventh cranial nerve. Mutant mice for Hoxb1 were reported to present with facial weakness, resembling MBS. Recently a homozygous mutation altering arg5 residue of HOXB1 homeodomain into cys5 was identified in two families with HCFP. We screened 95 sporadic patients diagnosed as MBS or HCFP for mutations in HOXB1. A novel homozygous alteration was identified in one HCFP case, affecting the same residue, resulting to his5. In silico protein analysis predicted stronger HOXB1-DNA binding properties for his5 than cys5 that resulted to milder phenotype. It should be noted that, inclusive of the previous report, only two mutations revealed in HOXB1 associated with HCFP involved the same amino acid arg5 in HOXB1 residing in HOXB1-DNA-PBX1 ternary complex.
Publicações recentes
Magnetic resonance imaging of developmental facial paresis: a spectrum of complex anomalies.
An exome sequencing study of Moebius syndrome including atypical cases reveals an individual with CFEOM3A and a TUBB3 mutation.
A novel homozygous HOXB1 mutation in a Turkish family with hereditary congenital facial paresis.
Homozygous HOXB1 loss-of-function mutation in a large family with hereditary congenital facial paresis.
A new hereditary congenital facial palsy case supports arg5 in HOX-DNA binding domain as possible hot spot for mutations.
📚 EuropePMCmostrando 5
Magnetic resonance imaging of developmental facial paresis: a spectrum of complex anomalies.
NeuroradiologyAn exome sequencing study of Moebius syndrome including atypical cases reveals an individual with CFEOM3A and a TUBB3 mutation.
Cold Spring Harbor molecular case studiesA novel homozygous HOXB1 mutation in a Turkish family with hereditary congenital facial paresis.
Brain & developmentHomozygous HOXB1 loss-of-function mutation in a large family with hereditary congenital facial paresis.
American journal of medical genetics. Part AA new hereditary congenital facial palsy case supports arg5 in HOX-DNA binding domain as possible hot spot for mutations.
European journal of medical geneticsAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Paralisia facial isolada hereditária congênita.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Paralisia facial isolada hereditária congênita
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Magnetic resonance imaging of developmental facial paresis: a spectrum of complex anomalies.
- A novel homozygous HOXB1 mutation in a Turkish family with hereditary congenital facial paresis.
- An exome sequencing study of Moebius syndrome including atypical cases reveals an individual with CFEOM3A and a TUBB3 mutation.
- Homozygous HOXB1 loss-of-function mutation in a large family with hereditary congenital facial paresis.
- A new hereditary congenital facial palsy case supports arg5 in HOX-DNA binding domain as possible hot spot for mutations.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:306527(Orphanet)
- MONDO:0011090(MONDO)
- GARD:8583(GARD (NIH))
- Busca completa no PubMed(PubMed)
- Q55783188(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar