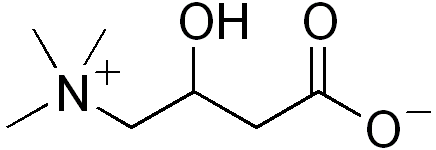
A deficiência de carnitina-acilcarnitina translocase (CACT) é uma doença genética grave e com risco de vida, que afeta a forma como o corpo usa gorduras para gerar energia. Ela costuma se manifestar logo após o nascimento, com sintomas como: hipoglicemia grave (nível muito baixo de açúcar no sangue) sem produção de energia alternativa; excesso de amônia no sangue; problemas no músculo do coração e/ou ritmo cardíaco irregular; mau funcionamento do fígado; fraqueza muscular generalizada; e alterações no cérebro.
Introdução
O que você precisa saber de cara
A deficiência de carnitina-acilcarnitina translocase (CACT) é uma doença genética grave e com risco de vida, que afeta a forma como o corpo usa gorduras para gerar energia. Ela costuma se manifestar logo após o nascimento, com sintomas como: hipoglicemia grave (nível muito baixo de açúcar no sangue) sem produção de energia alternativa; excesso de amônia no sangue; problemas no músculo do coração e/ou ritmo cardíaco irregular; mau funcionamento do fígado; fraqueza muscular generalizada; e alterações no cérebro.
Escala de raridade
<1/50kMuito rara
1/20kRara
1/10kPouco freq.
1/5kIncomum
1/2k
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Partes do corpo afetadas
+ 17 sintomas em outras categorias
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 45 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Triagem neonatal (Teste do Pezinho)
A triagem neonatal permite diagnóstico precoce e início imediato do tratamento.
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Genes associados
1 gene identificado com associação a esta condição. Padrão de herança: Autosomal recessive.
Mediates the electroneutral exchange of acylcarnitines (O-acyl-(R)-carnitine or L-acylcarnitine) of different acyl chain lengths (ranging from O-acetyl-(R)-carnitine to long-chain O-acyl-(R)-carnitines) with free carnitine ((R)-carnitine or L-carnitine) across the mitochondrial inner membrane, via a ping-pong mechanism (Probable) (PubMed:12892634, PubMed:18307102). Key player in the mitochondrial oxidation pathway, it translocates the fatty acids in the form of acylcarnitines into the mitochondr
Mitochondrion inner membrane
Carnitine-acylcarnitine translocase deficiency
A rare long-chain fatty acid oxidation disorder. Metabolic consequences include hypoketotic hypoglycemia under fasting conditions, hyperammonemia, elevated creatine kinase and transaminases, dicarboxylic aciduria, very low free carnitine and abnormal acylcarnitine profile with marked elevation of the long-chain acylcarnitines. Clinical features include neurologic abnormalities, cardiomyopathy, arrhythmias, skeletal muscle damage, liver dysfunction and episodes of life-threatening coma, which eventually lead to death. Most patients become symptomatic in the neonatal period with a rapidly progressive deterioration and a high mortality rate.
Variantes genéticas (ClinVar)
117 variantes patogênicas registradas no ClinVar.
Classificação de variantes (ClinVar)
Distribuição de 308 variantes classificadas pelo ClinVar.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Deficiência de carnitina-acilcarnitina translocase
Selecione um estado ou use sua localização para ver resultados.
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Ensaios em destaque
🟢 Recrutando agora
1 pesquisa recrutando participantes. Converse com seu médico sobre a possibilidade de participar.
Outros ensaios clínicos
9 ensaios clínicos encontrados, 1 ativos.
Publicações mais relevantes
Successful Management by Selective Embryo in the Carnitine-acylcarnitine Translocase Deficiency with SLC25A20 C.199-10T>G Variation: The First Case Report from Vietnam and Literature Review.
Carnitine-acylcarnitine translocase deficiency with SLC25A20 c.199-10T>G variation is a rare condition, typically associated with severe neonatal outcomes. Recently, preimplantation genetic testing (PGT) has emerged as a screening test applicable to embryos produced through in vitro fertilization for genetic analysis before transfer. Thus, PGT allows for the identification and elimination of embryos carrying inherited genetic diseases. This case report aims to present data from PGT on intervention in the management of SLC25A20 c.199-10T>G variation, particularly in middle-income countries. A 26-year-old woman with a high-risk term pregnancy and a history of two sudden neonatal deaths underwent parental carrier testing, revealing heterozygous SLC25A20 c.199-10T>G variation in both parents. The subsequent pregnancy, identified as a homozygous for SLC25A20 c.199-10T>G mutation, was terminated at 20 weeks. The current pregnancy was successfully managed by in vitro fertilization-selective embryo transfer. Carnitine-acylcarnitine translocase deficiency owing to SLC25A20 c.199-10T>G variation can result in sudden neonatal collapse. Obstetricians should maintain a high index of suspicion in recurrent cases of unexplained early neonatal death. Parental carrier testing is crucial for prenatal management, and selective embryo transfer is a core treatment for heterozygous SLC25A20 gene carriers in this highly lethal disorder.
Palmitoyl-carnitine Regulates Lung Development by Promoting Pulmonary Mesenchyme Proliferation.
Disruption of acylcarnitine homeostasis results in life-threatening outcomes in humans. Carnitine-acylcarnitine translocase deficiency (CACTD) is a scarce autosomal recessive genetic disease and may result in patients' death due to heart arrest or respiratory insufficiency. However, the reasons and mechanism of CACTD inducing respiratory insufficiency have never been elucidated. Herein, we employed lipidomic techniques to create comprehensive lipidomic maps of entire lungs throughout both prenatal and postnatal developmental stages in mice. We found that the acylcarnitines manifested notable variations and coordinated the expression levels of carnitine-acylcarnitine translocase (Cact) across these lung developmental stages. Cact-null mice were all dead with a symptom of respiratory distress and exhibited failed lung development. Loss of Cact resulted in an accumulation of palmitoyl-carnitine (C16-acylcarnitine) in the lungs and promoted the proliferation of mesenchymal progenitor cells. Mesenchymal cells with elevated C16-acylcarnitine levels displayed minimal changes in energy metabolism but, upon investigation, revealed an interaction with sterile alpha motif domain and histidine-aspartate domain-containing protein 1 (Samhd1), leading to decreased protein abundance and enhanced cell proliferation. Thus, our findings present a mechanism addressing respiratory distress in CACTD, offering a valuable reference point for both the elucidation of pathogenesis and the exploration of treatment strategies for neonatal respiratory distress.
Carglumic acid as a treatment for persistent hyperammonemia in carnitine-acylcarnitine translocase deficiency: A case study.
Carnitine-acylcarnitine translocase deficiency (CACTD) is a rare autosomal recessive fatty acid oxidation disorder resulting in energy deficiency due to impaired mitochondrial long-chain fatty acid transport. Hyperammonemia is a critical complication, often resistant to conventional treatment. Here, we report the case of a 7-month-old patient with CACTD, initially diagnosed at 10 days old, who presented with persistent hyperammonemia despite optimized medical nutrition therapy and conventional nitrogen scavenging with sodium benzoate. When hyperammonemia persisted, carglumic acid was introduced, leading to a sustained decrease in ammonia levels and effective long-term control. Carglumic acid, typically indicated for organic acidemias, proved beneficial in this CACTD case. The administration of carglumic acid not only provided acute resolution but also stabilized ammonia levels over prolonged follow-up. This case highlights carglumic acid as a potential therapeutic option for managing hyperammonemia in CACTD, underscoring the need for further studies to confirm its efficacy in long-term management of hyperammonemia in fatty acid oxidation disorders.
Loss-of-function SLC25A20 variant causes carnitine-acylcarnitine translocase deficiency by reducing SLC25A20 protein stability.
Autosomal-recessive carnitine-acylcarnitine translocase deficiency (CACTD) is a rare disorder of long-chain fatty acid oxidation caused by variants in the SLC25A20 gene, leading to energy deficiency and the toxic accumulation of long-chain acylcarnitines. Under fasting conditions, most newborns with severe CACTD experience sudden cardiac arrest and hypotonia, often leading to premature death due to rapid disease progression. The genetic factors and pathogenic mechanisms in CACTD are essential for its diagnosis, treatment, and prevention. Whole-exome sequencing was carried out on the CACTD patients. Bioinformatics analysis predicted the pathogenicity and three-dimensional structure of SLC25A20. Quantitative PCR was employed to detect changes in SLC25A20, CPT1A and CPT2 mRNA levels. The expression and stability of the variant protein were assessed via Western blot. Additionally, the subcellular localization of the variant protein was observed using immunofluorescence. We identified compound heterozygous pathogenic variants of SLC25A20 (c.476 T > C and c.199-10 T > G) in CACTD families, with patients exhibiting an abnormal carnitine spectrum. In vitro functional studies demonstrated that the c.476 T > C and c.199-10 T > G variants decreased the protein stability of SLC25A20, reduced CPT1A and CPT2 mRNA expression, and caused protein aggregation of SLC25A20. We propose that the decreased stability of the SLC25A20 variants c.476 T > C and c.199-10 T > G has the potential to lead to the development of CACTD by affecting the mitochondrial shuttle of acylcarnitine and carnitine, thereby inhibiting the β-oxidation pathway. Therefore, we believe these compound heterozygous variants (c.199-10 T > G and c.476 T > C) are loss-of-function variants. Our findings provide valuable data on CACTD pathogenesis and genotype-phenotype correlations.
Homozygous slc25a20 zebrafish mutant reveals insights into carnitine-acylcarnitine translocase deficiency pathogenesis.
The SLC25A20 gene encodes carnitine-acylcarnitine translocase (CACT), facilitating the transport of long-chain acylcarnitine required for energy production via β-oxidation into the mitochondria. Loss-of-function mutations in this gene lead to CACT deficiency, a rare autosomal recessive disorder of fatty acid metabolism characterized by severe symptoms including cardiomyopathy, hepatic dysfunction, rhabdomyolysis, hypoketotic hypoglycemia, and hyperammonemia, often resulting in neonatal mortality. Here, we utilized CRISPR/Cas9 gene editing to isolate slc25a20 mutant zebrafish. Homozygous mutants displayed significant lethality, with the majority succumbing before reaching maturity. However, we identified a notably rare homozygous individual that survived into adulthood, prompting a histological examination. Firstly, we observed adipose tissue accumulation at various sites in the homozygous mutant. The mutant heart exhibited hypertrophy, along with degenerated myocardial and muscle cells containing numerous eosinophilic nuclei. Additionally, we found no large oil droplet vacuoles in the mutant liver; however, the hepatocytes displayed numerous small vacuoles resembling lipid droplets. Iron deposition was evident in the spleen and parts of the liver. Overall, our slc25a20 zebrafish mutant displayed tissue pathologies analogous to human CACT deficiency, suggesting its potential as a pathological model contributing to the elucidation of pathogenesis and the improvement/development of therapies for CACT deficiency.
Publicações recentes
Successful Management by Selective Embryo in the Carnitine-acylcarnitine Translocase Deficiency with SLC25A20 C.199-10T>G Variation: The First Case Report from Vietnam and Literature Review.
Palmitoyl-carnitine Regulates Lung Development by Promoting Pulmonary Mesenchyme Proliferation.
Carglumic acid as a treatment for persistent hyperammonemia in carnitine-acylcarnitine translocase deficiency: A case study.
🥇 Revisão sistemáticaLoss-of-function SLC25A20 variant causes carnitine-acylcarnitine translocase deficiency by reducing SLC25A20 protein stability.
Homozygous slc25a20 zebrafish mutant reveals insights into carnitine-acylcarnitine translocase deficiency pathogenesis.
📚 EuropePMC54 artigos no totalmostrando 40
Successful Management by Selective Embryo in the Carnitine-acylcarnitine Translocase Deficiency with SLC25A20 C.199-10T>G Variation: The First Case Report from Vietnam and Literature Review.
Oman medical journalPalmitoyl-carnitine Regulates Lung Development by Promoting Pulmonary Mesenchyme Proliferation.
Research (Washington, D.C.)Carglumic acid as a treatment for persistent hyperammonemia in carnitine-acylcarnitine translocase deficiency: A case study.
Molecular genetics and metabolism reportsLoss-of-function SLC25A20 variant causes carnitine-acylcarnitine translocase deficiency by reducing SLC25A20 protein stability.
GeneHomozygous slc25a20 zebrafish mutant reveals insights into carnitine-acylcarnitine translocase deficiency pathogenesis.
Molecular genetics and metabolism reportsCarnitine-acylcarnitine translocase deficiency: a case report with autopsy.
Autopsy & case reports[Clinical and genetic analysis of two pedigrees affected with Carnitine-acylcarnitine translocase deficiency due to variant of SLC25A20 gene].
Zhonghua yi xue yi chuan xue za zhi = Zhonghua yixue yichuanxue zazhi = Chinese journal of medical geneticsSudden death with cardiac involvement in a neonate with carnitine-acylcarnitine translocase deficiency.
Cardiovascular pathology : the official journal of the Society for Cardiovascular PathologyThe significance of acylcarnitine ratio indices in diagnosing carnitine-acylcarnitine translocase deficiency.
Translational pediatricsAcylcarnitine ratio indices in diagnosing carnitine-acylcarnitine translocase deficiency in newborns.
Translational pediatricsIncreased acylcarnitine ratio indices in newborn screening for carnitine-acylcarnitine translocase deficiency shows increased sensitivity and reduced false-positivity.
Translational pediatricsCarnitine-acylcarnitine translocase deficiency caused by SLC25A20 gene heterozygous variants in twins: a case report.
The Journal of international medical researchDataset from dried blood spot acylcarnitine for detection of Carnitine-Acylcarnitine Translocase (CACT) deficiency and Carnitine Palmitoyl Transferase 2 (CPT2) deficiency.
Data in briefIn Silico Analysis of the Structural Dynamics and Substrate Recognition Determinants of the Human Mitochondrial Carnitine/Acylcarnitine SLC25A20 Transporter.
International journal of molecular sciencesCarnitine-acylcarnitine Translocase Deficiency with c.199-10T>G Mutation in Two Filipino Neonates Detected through Parental Carrier Testing.
International journal of neonatal screeningOne potential hotspot SLC25A20 gene variants in Chinese patients with carnitine-acylcarnitine translocase deficiency.
Frontiers in pediatricsNewborn Screening for Mitochondrial Carnitine-Acylcarnitine Cycle Disorders in Zhejiang Province, China.
Frontiers in geneticsWhole exome sequencing analysis in a couple with three children who died prematurely due to carnitine-acylcarnitine translocase deficiency.
Taiwanese journal of obstetrics & gynecologyNeonatal sudden death caused by a novel heterozygous mutation in SLC25A20 gene: A case report and brief literature review.
Legal medicine (Tokyo, Japan)Screening for neonatal inherited metabolic disorders by tandem mass spectrometry in Guangzhou.
Zhejiang da xue xue bao. Yi xue ban = Journal of Zhejiang University. Medical sciencesNovel mutations associated with carnitine-acylcarnitine translocase and carnitine palmitoyl transferase 2 deficiencies in Malaysia.
Clinical biochemistryClinical and molecular characteristics of carnitineacylcarnitine translocase deficiency with c.270delC and a novel c.408C>A variant.
The Turkish journal of pediatricsNew insights into carnitine-acylcarnitine translocase deficiency from 23 cases: Management challenges and potential therapeutic approaches.
Journal of inherited metabolic diseaseCarnitine-Acylcarnitine Translocase Deficiency Masked by Extreme Prematurity.
Journal of pediatric geneticsLate-Onset Carnitine-Acylcarnitine Translocase Deficiency With SLC25A20 c.199-10T>G Variation: Case Report and Pathologic Analysis of Liver Biopsy.
Frontiers in pediatricsTutorial: Triheptanoin and Nutrition Management for Treatment of Long-Chain Fatty Acid Oxidation Disorders.
JPEN. Journal of parenteral and enteral nutritionDiagnosis, genetic characterization and clinical follow up of mitochondrial fatty acid oxidation disorders in the new era of expanded newborn screening: A single centre experience.
Molecular genetics and metabolism reportsThe use of sodium DL-3-Hydroxybutyrate in severe acute neuro-metabolic compromise in patients with inherited ketone body synthetic disorders.
Orphanet journal of rare diseasesDiagnostic contribution of metabolic workup for neonatal inherited metabolic disorders in the absence of expanded newborn screening.
Scientific reportsEpidemiology of rare diseases detected by newborn screening in the Czech Republic.
Central European journal of public healthClinical and molecular characteristics of carnitine-acylcarnitine translocase deficiency: Experience with six patients in Guangdong China.
Clinica chimica acta; international journal of clinical chemistry[Analysis of four carnitine-acylcarnitine translocase deficiency cases caused by homozygous mutation of SLC25A20 c.199-10T> G].
Zhonghua er ke za zhi = Chinese journal of pediatricsThe safety of Lipistart, a medium-chain triglyceride based formula, in the dietary treatment of long-chain fatty acid disorders: a phase I study.
Journal of pediatric endocrinology & metabolism : JPEMCarnitine-acylcarnitine translocase deficiency with c.199-10 T>G and novel c.1A>G mutation: Two case reports and brief literature review.
MedicineExpanded newborn metabolic screening programme in Hong Kong: a three-year journey.
Hong Kong medical journal = Xianggang yi xue za zhiSudden infant death from neonate carnitine palmitoyl transferase II deficiency.
Forensic science internationalTriheptanoin: A Rescue Therapy for Cardiogenic Shock in Carnitine-acylcarnitine Translocase Deficiency.
JIMD reportsHistorical Perspective on Clinical Trials of Carnitine in Children and Adults.
Annals of nutrition & metabolismA novel method for determining peroxisomal fatty acid β-oxidation.
Journal of inherited metabolic diseaseCarnitine-acylcarnitine translocase deficiency: experience with four cases in Spain and review of the literature.
JIMD reportsAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Deficiência de carnitina-acilcarnitina translocase.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Deficiência de carnitina-acilcarnitina translocase
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Successful Management by Selective Embryo in the Carnitine-acylcarnitine Translocase Deficiency with SLC25A20 C.199-10T>G Variation: The First Case Report from Vietnam and Literature Review.
- Palmitoyl-carnitine Regulates Lung Development by Promoting Pulmonary Mesenchyme Proliferation.
- Carglumic acid as a treatment for persistent hyperammonemia in carnitine-acylcarnitine translocase deficiency: A case study.
- Loss-of-function SLC25A20 variant causes carnitine-acylcarnitine translocase deficiency by reducing SLC25A20 protein stability.
- Homozygous slc25a20 zebrafish mutant reveals insights into carnitine-acylcarnitine translocase deficiency pathogenesis.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:159(Orphanet)
- OMIM OMIM:212138(OMIM)
- MONDO:0008918(MONDO)
- GARD:1123(GARD (NIH))
- Variantes catalogadas(ClinVar)
- Busca completa no PubMed(PubMed)
- Q5044061(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar