A deficiência da LCAT (enzima lecitina-colesterol aciltransferase) é um problema raro no metabolismo das lipoproteínas, que se manifesta com opacidades na córnea (manchas esbranquiçadas nos olhos), e às vezes com insuficiência renal e anemia hemolítica. Em exames de laboratório, é caracterizada por níveis muito baixos do colesterol HDL (o "colesterol bom").
Introdução
O que você precisa saber de cara
A deficiência da LCAT (enzima lecitina-colesterol aciltransferase) é um problema raro no metabolismo das lipoproteínas, que se manifesta com opacidades na córnea (manchas esbranquiçadas nos olhos), e às vezes com insuficiência renal e anemia hemolítica. Em exames de laboratório, é caracterizada por níveis muito baixos do colesterol HDL (o "colesterol bom").
Escala de raridade
<1/50kMuito rara
1/20kRara
1/10kPouco freq.
1/5kIncomum
1/2k
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Partes do corpo afetadas
+ 8 sintomas em outras categorias
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 24 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Genes associados
1 gene identificado com associação a esta condição. Padrão de herança: Autosomal recessive.
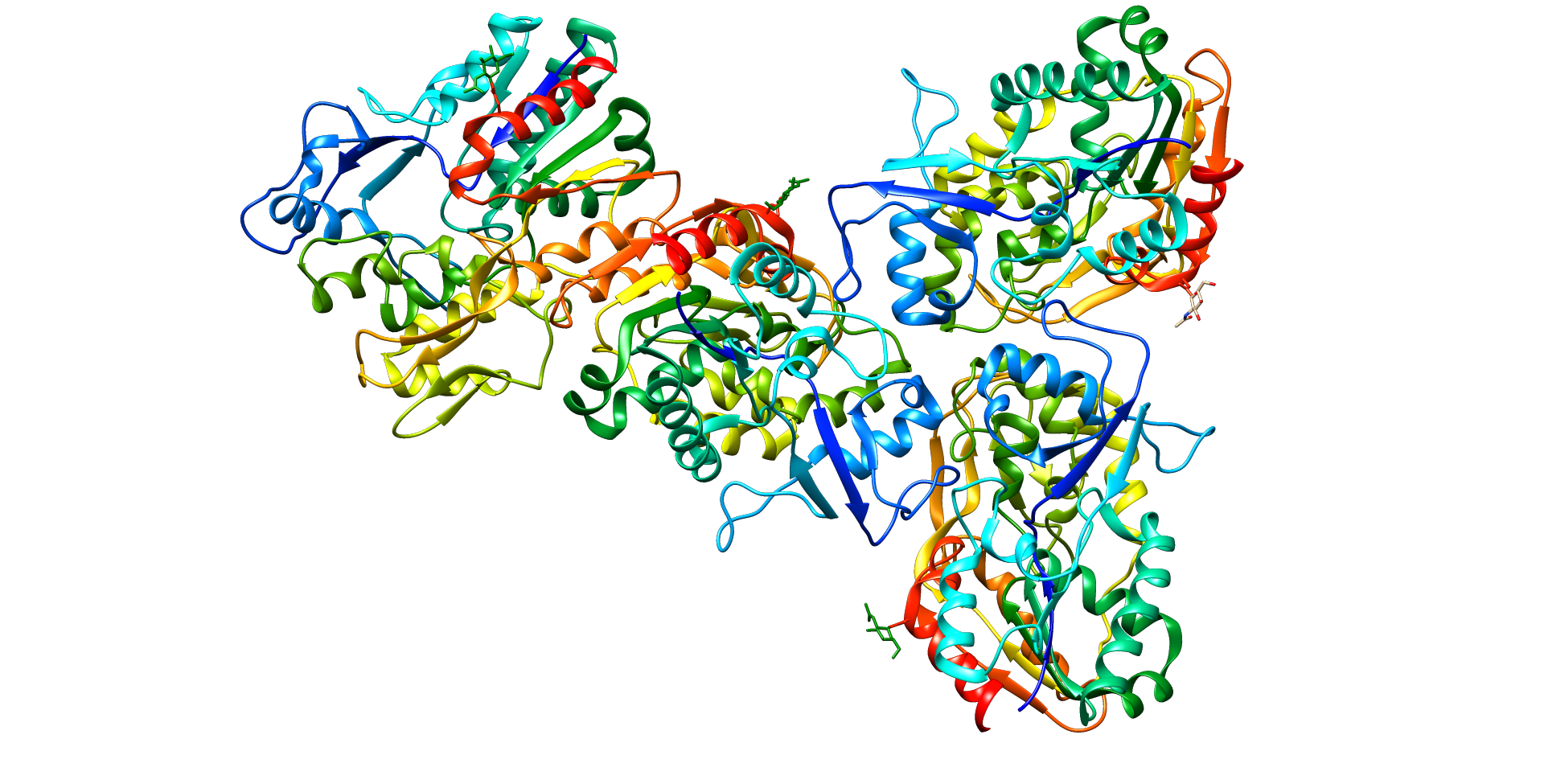
Central enzyme in the extracellular metabolism of plasma lipoproteins. Synthesized mainly in the liver and secreted into plasma where it converts cholesterol and phosphatidylcholines (lecithins) to cholesteryl esters and lysophosphatidylcholines on the surface of high and low density lipoproteins (HDLs and LDLs) (PubMed:10329423, PubMed:19065001, PubMed:26195816). The cholesterol ester is then transported back to the liver. Has a preference for plasma 16:0-18:2 or 18:O-18:2 phosphatidylcholines
Secreted
Lecithin-cholesterol acyltransferase deficiency
A disorder of lipoprotein metabolism characterized by inadequate esterification of plasmatic cholesterol. Two clinical forms are recognized: complete LCAT deficiency and fish-eye disease. LCATD is generally referred to the complete form which is associated with absence of both alpha and beta LCAT activities resulting in esterification anomalies involving both HDL (alpha-LCAT activity) and LDL (beta-LCAT activity). It causes a typical triad of diffuse corneal opacities, target cell hemolytic anemia, and proteinuria with renal failure.
Variantes genéticas (ClinVar)
77 variantes patogênicas registradas no ClinVar.
Classificação de variantes (ClinVar)
Distribuição de 38 variantes classificadas pelo ClinVar.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Deficiência de LCAT
Selecione um estado ou use sua localização para ver resultados.
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Ensaios em destaque
🟢 Recrutando agora
1 pesquisa recrutando participantes. Converse com seu médico sobre a possibilidade de participar.
Outros ensaios clínicos
60 ensaios clínicos encontrados, 1 ativos.
Publicações mais relevantes
Lecithin-cholesterol acyltransferase deficiency with the finding of "zebra bodies": A diagnostic challenge in the context of suspected Fabry disease.
Lecithin-cholesterol acyltransferase (LCAT) deficiency is a rare autosomal recessive disorder resulting from mutations in the LCAT gene, which leads to abnormal lipoprotein metabolism. This results in markedly reduced high-density lipoprotein cholesterol and the accumulation of lipoprotein X, leading to renal, corneal, and hemolytic damage. Two clinical variants have been described: familial LCAT deficiency (FLD) and fish-eye disease (FED). We report the case of a 41-year-old male with a history of hypertension, tinnitus, and progressive hearing loss, who presented with bilateral corneal opacity since childhood. Laboratory studies revealed significant proteinuria (2.56 g/24 h), preserved renal function (creatinine 0.85 mg/dL), mild anemia (Hb 10.2 g/dL), and extremely low HDL cholesterol (1.3 mg/dL). Renal biopsy showed focal segmental glomerulosclerosis and mesangial expansion. Electron microscopy demonstrated concentric lamellar inclusions known as "zebra bodies," a finding typically associated with Fabry disease. However, α-galactosidase A activity was normal, and genetic testing for Fabry disease was negative. Further genetic analysis identified the variant c.757 p.(Gln253Argfs*11) in the LCAT gene, confirming the diagnosis of familial LCAT deficiency. This case highlights the importance of differentiating LCAT deficiency from Fabry disease, given their overlapping clinical and histological features. Moreover, it represents the first description of "zebra bodies" in LCAT deficiency, emphasizing the diagnostic complexity and the need for a multidisciplinary approach to ensure accurate diagnosis and appropriate management.
Lecithin-Cholesterol Acyltransferase Deficiency as a Rare Cause of Bilateral Corneal Opacities: A Case Report of a Novel Frameshift Mutation.
Lecithin-cholesterol acyltransferase (LCAT) deficiency is a rare autosomal recessive disorder of lipid metabolism characterized by corneal opacification, hemolytic anemia, and chronic kidney disease. We describe the ophthalmic, systemic, and genetic findings of a patient with LCAT deficiency and report a novel frameshift mutation in the LCAT gene. Ophthalmic findings may represent the first clinical sign and guide the diagnosis. A 50-year-old white male with end-stage renal disease on hemodialysis and a history of recurrent hemolytic anemia was referred for bilateral corneal opacities. Despite diffuse opacification involving all corneal layers, his best corrected visual acuity remained 20/20 in both eyes with normal color vision, although contrast sensitivity was reduced. Laboratory testing revealed normocytic, normochromic anemia, low HDL cholesterol, and reduced apolipoprotein A levels. Genetic analysis identified compound heterozygosity in the LCAT gene: a novel frameshift variant c.580_598del p.(Ala194Serfs*64), classified as likely pathogenic, and the previously described missense variant c.619G>A p.(Gly207Ser), also classified as likely pathogenic. This case highlights the importance of considering metabolic disorders in the differential diagnosis of bilateral corneal opacities and expands the genetic spectrum of LCAT deficiency by reporting a novel frameshift mutation.
A Case of Acquired LCAT Deficiency with the Discrepancy between Spontaneous Resolution of Proteinuria and Continually Low HDL Cholesterol Levels.
A 79-year-old Chinese man was referred for nephrotic syndrome (proteinuria 4.4 g/day). In blood tests, serum high-density lipoprotein (HDL) cholesterol was undetectable, and the esterified cholesterol to total cholesterol ratio was very low. Lecithin: cholesterol acyltransferase (LCAT) activity was also undetectable. Since he had neither corneal opacity nor pathological mutations in the LCAT gene and anti-LCAT antibodies were detected in serum, a diagnosis of acquired LCAT deficiency was made. Renal biopsy revealed glomerulopathy associated with LCAT deficiency and membranous nephropathy (MN). Since the patient's proteinuria did not improve despite prescribing an angiotensin II receptor blocker (ARB), we suggested the prescription of prednisolone, but he returned to China due to the expiration of his residence visa for Japan. One year after the initial visit, his proteinuria had improved to 0.9 g/day without immunosuppressive therapy. However, his HDL cholesterol level was still low at around 3 mg/dL, indicating a discrepancy between remission of nephrotic syndrome and lack of improvement in lipid levels.Of the 11 patients with acquired LCAT deficiency reported to date, 4 with undetectable LCAT activity and MN on renal biopsy required immunosuppressive therapy to alleviate proteinuria. The present patient was prescribed only an ARB according to his preference, which happened to be consistent with the MN treatment guideline that states, "Wait 6 months for spontaneous remission while using maximal antiproteinuric therapy." The clinical course of acquired LCAT deficiency varies, and further case reports are needed to determine the necessity of immunosuppressive therapy.
Loss of LCAT function aggravates metabolic-associated steatohepatitis (MASH) in golden Syrian hamster.
Lecithin cholesterol acyltransferase (LCAT) plays a pivotal role in acyl-esterifying cholesterol intravascularly, but its function in metabolic dysfunction-associated steatotic liver disease (MASLD) or steatohepatitis (MASH) has remained uncertain both in murine models and humans for decades, which is largely attributable to the distinct differences in cholesterol metabolism between mice and humans. Previously, we created a novel golden Syrian hamster model deficient in LCAT activity. Herein, we explored the influence of LCAT on the development of MASLD and MASH. A cross-sectional clinical study of LCAT activity and free cholesterol (FC) levels in healthy and MASLD patients was performed. LCAT knockout (LCAT KO) hamsters were used to explore the characteristics of cholesterol homeostasis and MASLD and MASH development. Lipidomics, mRNA-seq, and qPCR were employed to investigate the underlying mechanisms involved. MASLD patients displayed reduced LCAT activity, elevated FC levels, and ratio of FC/TC. Serum FC levels were positively correlated with triglyceride (TG), total cholesterol (TC), and apoB100 levels. In hamsters, LCAT deficiency resulted in increased FC levels and decreased high-density lipoprotein levels. Apolipoprotein profiles revealed increased ApoB100/48 and apoE but decreased apoAI. Increases in serum FC levels were primarily observed in LCAT-deficient hamster. Interestingly, LCAT KO hamsters presented mild TG species deposition in the liver even when fed a chow diet indicated by lipidomics. These increased TG species included TG (16:0/18:1/18:2), TG (16:0/18:1/18:3), and TG (16:0/16:1/18:1). On a high-fat and high-cholesterol diet, LCAT-deficient hamsters developed severe liver ballooning, inflammation, and fibrosis. Using HepG2 cells and primary hepatocytes confirmed that FC increased intracellular lipogenesis and promoted inflammatory response, which was reversed by a NLRP3 inhibitor. In summary, LCAT deficiency in hamsters promotes liver lipid deposition and MASH progression, thus highlighting the therapeutic role of LCAT in MASLD and MASH.
Quantitative Assessment of Visual Function in Japanese Patients With Lecithin-Cholesterol Acyltransferase Gene Abnormalities: A Case-Control Study.
Introduction Fish-eye disease (FED) and familial lecithin-cholesterol acyltransferase (LCAT) deficiency (FLD) are rare. The aim of this study was to compare visual function between patients with LCAT abnormalities - namely, FED and FLD - and healthy controls. Methods This retrospective, comparative case-control study included four patients with FLD or FED (LCAT group) who presented with cloudy corneas at Miyata Eye Hospital between 2018 and 2024. Four age- and sex-matched individuals with normal results on ophthalmic examination were included as controls. We reviewed medical records for best-corrected visual acuity (BCVA), corneal astigmatism, forward light scattering, and contrast sensitivity. The parameters were compared between the groups. Results Sixteen eyes of eight women were included, including eight eyes of four patients with cloudy corneas in the LCAT group (two with FLD and six with FED) and eight eyes of four controls. The mean BCVA and corneal astigmatism revealed no significant intergroup differences. However, forward scattering was significantly higher in the LCAT group than in the control group (p = 0.007). The area under the log-contrast sensitivity function was significantly lower in the LCAT group than in the control group (p = 0.017). Conclusions Despite normal BCVA, patients with LCAT abnormalities (FLD and FED) showed considerably increased forward light scattering and decreased contrast sensitivity compared with the controls, indicating subtle but substantial visual functional impairment.
Publicações recentes
Lecithin-cholesterol acyltransferase deficiency with the finding of "zebra bodies": A diagnostic challenge in the context of suspected Fabry disease.
Lecithin-Cholesterol Acyltransferase Deficiency as a Rare Cause of Bilateral Corneal Opacities: A Case Report of a Novel Frameshift Mutation.
Quantitative Assessment of Visual Function in Japanese Patients With Lecithin-Cholesterol Acyltransferase Gene Abnormalities: A Case-Control Study.
Loss of LCAT function aggravates metabolic-associated steatohepatitis (MASH) in golden Syrian hamster.
IgG3κ Monoclonal Membranous Nephropathy Associated With Acquired Lecithin Cholesterol Acyltransferase Deficiency.
📚 EuropePMC123 artigos no totalmostrando 98
Lecithin-cholesterol acyltransferase deficiency with the finding of "zebra bodies": A diagnostic challenge in the context of suspected Fabry disease.
NefrologiaLecithin-Cholesterol Acyltransferase Deficiency as a Rare Cause of Bilateral Corneal Opacities: A Case Report of a Novel Frameshift Mutation.
Case reports in ophthalmologyQuantitative Assessment of Visual Function in Japanese Patients With Lecithin-Cholesterol Acyltransferase Gene Abnormalities: A Case-Control Study.
CureusLoss of LCAT function aggravates metabolic-associated steatohepatitis (MASH) in golden Syrian hamster.
Clinical science (London, England : 1979)IgG3κ Monoclonal Membranous Nephropathy Associated With Acquired Lecithin Cholesterol Acyltransferase Deficiency.
American journal of kidney diseases : the official journal of the National Kidney FoundationA Case of Acquired LCAT Deficiency with the Discrepancy between Spontaneous Resolution of Proteinuria and Continually Low HDL Cholesterol Levels.
Journal of atherosclerosis and thrombosisShort-Term Treatment for Immune-Mediated Acquired Lecithin-Cholesterol Acyltransferase Deficiency Restores the High-Density Lipoprotein Function: A Case Report.
Journal of atherosclerosis and thrombosisLipoprotein X - Pathophysiology, diagnosis, and management.
Journal of clinical lipidologyLCAT deficiency promotes hepatocellular carcinoma progression and lenvatinib resistance by promoting triglyceride catabolism and fatty acid oxidation.
Cancer lettersNovel pathogenic variant in the LCAT gene in a compound heterozygous patient with fish-eye disease and a mild phenotype.
Journal of clinical lipidologyGliflozins, sucrose and flavonoids are allosteric activators of lecithin-cholesterol acyltransferase.
Scientific reportsA Novel Symptomatic Lecithin-Cholesterol Acyltransferase Gene Mutation With Corneal Amyloidosis.
CorneaRescue of Familial Lecithin:Cholesterol Acyltranferase Deficiency Mutations with an Allosteric Activator.
Molecular pharmacologyUnveiling Renal Lipid Deposition: A Rare Case of Hepatic Glomerulosclerosis Resembling Lecithin-Cholesterol Acyltransferase (LCAT) Deficiency Post Liver Transplantation.
CureusA novel splicing variant in ABCA1 in the first reported Hong Kong Chinese patient with high-density lipoprotein deficiency.
Endocrinology, diabetes & metabolism case reportsLongitudinal analysis of clinical and laboratory biomarkers in a patient with familial lecithin: cholesterol acyltransferase deficiency (FLD) and accelerated eGFR decline: A case study.
Journal of clinical lipidologyEstrogen Induces LCAT to Maintain Cholesterol Homeostasis and Suppress Hepatocellular Carcinoma Development.
Cancer researchFamilial LCAT Deficiency and Low HDL-C Levels: In silico Characterization of Two Rare LCAT Missense Mutations.
The application of clinical geneticsA High-Throughput NMR Method for Lipoprotein-X Quantification.
Molecules (Basel, Switzerland)Abnormal Lipoproteins Trigger Oxidative Stress-Mediated Apoptosis of Renal Cells in LCAT Deficiency.
Antioxidants (Basel, Switzerland)HDL and chronic kidney disease.
Atherosclerosis plusVery low HDL levels: clinical assessment and management.
Archives of endocrinology and metabolismTwo novel variants in the lecithin:cholesterol acyltransferase gene resulted in classic LCAT deficiency.
Atherosclerosis plusA rare case of nephrotic syndrome and Tangier disease.
CEN case reportsNovel therapeutic opportunities for familial lecithin:cholesterol acyltransferase deficiency: promises and challenges.
Current opinion in lipidologyRapidly progressive renal failure to reveal LCAT deficiency in an Algerian family.
Clinical nephrologyFirst-in-human autologous implantation of genetically modified adipocytes expressing LCAT for the treatment of familial LCAT deficiency.
HeliyonApolipoprotein-A-I for severe COVID-19-induced hyperinflammatory states: A prospective case study.
Frontiers in pharmacologyA rare case of renal involvement in Lecithin-Cholesterol Acyltransferase (LCAT) deficiency: lessons for the clinical nephrologist.
Journal of nephrologyA Simple Fluorescent Cholesterol Labeling Method to Cryoprotect and Detect Plasma Lipoprotein-X.
BiologyLCAT-trial-24 weeks: Protocol for a clinical study to evaluate the safety of regenerative medicine and gene therapy by the autologous transplantation of human lecithin:cholesterol acyltransferase gene-transduced human pre-adipocytes.
Contemporary clinical trials communicationsPlasma FA composition in familial LCAT deficiency indicates SOAT2-derived cholesteryl ester formation in humans.
Journal of lipid researchFamilial lecithin-cholesterol acyltransferase deficiency: If so rare, why so frequent in the state of Piauí, northeastern Brazil?
Molecular genetics and metabolism reportsClinical Dilemma of Corneal Opacity, Very Low High-density Lipoprotein, and Nephrotic Syndrome: Mystery Revealed.
Saudi journal of kidney diseases and transplantation : an official publication of the Saudi Center for Organ Transplantation, Saudi ArabiaLCAT- targeted therapies: Progress, failures and future.
Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapieA systematic review of the natural history and biomarkers of primary lecithin:cholesterol acyltransferase deficiency.
Journal of lipid researchA novel pathogenic variant in LCAT causing FLD. A case report.
Acta clinica BelgicaThe HDL mimetic CER-001 remodels plasma lipoproteins and reduces kidney lipid deposits in inherited lecithin:cholesterol acyltransferase deficiency.
Journal of internal medicineLCAT deficiency and pregnancy: Case report.
Obstetric medicineLCAT deficiency: a systematic review with the clinical and genetic description of Mexican kindred.
Lipids in health and diseaseUsing Literature Based Discovery to Gain Insights Into the Metabolomic Processes of Cardiac Arrest.
Frontiers in research metrics and analyticsCurrent Status of Familial LCAT Deficiency in Japan.
Journal of atherosclerosis and thrombosisHigh-Density Lipoproteins and the Kidney.
CellsrHDL modeling and the anchoring mechanism of LCAT activation.
Journal of lipid researchCER-001 ameliorates lipid profile and kidney disease in a mouse model of familial LCAT deficiency.
Metabolism: clinical and experimentalProgression of chronic kidney disease in familial LCAT deficiency: a follow-up of the Italian cohort.
Journal of lipid researchSpontaneous Atherosclerosis in Aged LCAT-Deficient Hamsters With Enhanced Oxidative Stress-Brief Report.
Arteriosclerosis, thrombosis, and vascular biologyActivation of Naturally Occurring Lecithin:Cholesterol Acyltransferase Mutants by a Novel Activator Compound.
The Journal of pharmacology and experimental therapeuticsGenetic, biochemical, and clinical features of LCAT deficiency: update for 2020.
Current opinion in lipidologyEsterification of 4β-hydroxycholesterol and other oxysterols in human plasma occurs independently of LCAT.
Journal of lipid researchFamilial LCAT deficiency and cardiovascular disease: the game is not over. A case of dramatic multivessel atherosclerosis.
Minerva medicaLecithin:cholesterol acyltransferase: symposium on 50 years of biomedical research from its discovery to latest findings.
Journal of lipid researchNovel lecithin: cholesterol acyltransferase-based therapeutic approaches.
Current opinion in lipidologyLCAT protects against Lipoprotein-X formation in a murine model of drug-induced intrahepatic cholestasis.
Pharmacology research & perspectivesLipid Profile Rather Than the LCAT Mutation Explains Renal Disease in Familial LCAT Deficiency.
Journal of clinical medicineIdentification and functional analysis of missense mutations in the lecithin cholesterol acyltransferase gene in a Chilean patient with hypoalphalipoproteinemia.
Lipids in health and diseaseThe P274S Mutation of Lecithin-Cholesterol Acyltransferase (LCAT) and Its Clinical Manifestations in a Large Kindred.
American journal of kidney diseases : the official journal of the National Kidney FoundationPlasma lipoprotein-X quantification on filipin-stained gels: monitoring recombinant LCAT treatment ex vivo.
Journal of lipid researchImpact of apolipoprotein A1- or lecithin:cholesterol acyltransferase-deficiency on white adipose tissue metabolic activity and glucose homeostasis in mice.
Biochimica et biophysica acta. Molecular basis of diseaseCase Report: Management of Corneal Clouding from Lecithin: Cholesterol Acyltransferase Deficiency.
Optometry and vision science : official publication of the American Academy of OptometryLCAT Enzyme Replacement Therapy Reduces LpX and Improves Kidney Function in a Mouse Model of Familial LCAT Deficiency.
The Journal of pharmacology and experimental therapeutics[Glomerulopathy associated with lecithin-cholesterol-acyltransferase deficiency: A case report and literature review].
Annales de pathologieA proteomic approach to identify novel disease biomarkers in LCAT deficiency.
Journal of proteomicsFinding a very rare mutation in non-Caucasian LCAT patients from Southwest Asia for the first time.
Journal of cellular biochemistryLipoprotein-X fifty years after its original discovery.
Nutrition, metabolism, and cardiovascular diseases : NMCDMolecular basis for activation of lecithin:cholesterol acyltransferase by a compound that increases HDL cholesterol.
eLifeOcular and Genetic Characteristics Observed in Two Cases of Fish-Eye Disease.
CorneaParadoxical fall in proteinuria during pregnancy in an LCAT-deficient patient-A case report.
Journal of clinical lipidologyLp8 is potentially associated with partial lecithin:cholesterol acyltransferase deficiency in a patient with primary biliary cirrhosis.
Journal of clinical lipidologyImmune-mediated acquired lecithin-cholesterol acyltransferase deficiency: A case report and literature review.
Journal of clinical lipidologyComplete and Partial Lecithin:Cholesterol Acyltransferase Deficiency Is Differentially Associated With Atherosclerosis.
CirculationLCAT deficiency as a cause of proteinuria and corneal opacification.
BMJ case reportsLoss of LCAT activity in the golden Syrian hamster elicits pro-atherogenic dyslipidemia and enhanced atherosclerosis.
Metabolism: clinical and experimentalNovel metabolic phenotypes in lecithin cholesterol acyltyransferase-deficient mice.
Current opinion in lipidologyNovel Missense LCAT Gene Mutation Associated with an Atypical Phenotype of Familial LCAT Deficiency in Two Portuguese Brothers.
JIMD reportsA novel homozygous mutation causing lecithin-cholesterol acyltransferase deficiency in a proband of Romanian origin with a record of extreme gestational hyperlipidemia.
Journal of clinical lipidologyDevelopment of a novel fluorescent activity assay for lecithin:cholesterol acyltransferase.
Annals of clinical biochemistryAJKD Atlas of Renal Pathology: Lecithin-Cholesterol Acyltransferase (LCAT) Deficiency.
American journal of kidney diseases : the official journal of the National Kidney FoundationA case of acquired lecithin:cholesterol acyltransferase deficiency with sarcoidosis that remitted spontaneously.
CEN case reportsDepletion in LpA-I:A-II particles enhances HDL-mediated endothelial protection in familial LCAT deficiency.
Journal of lipid research[Lecithin:Cholesterol Acyltransferase Deficiency, from genes to therapy].
Giornale italiano di nefrologia : organo ufficiale della Societa italiana di nefrologiaFamilial Lecithin Cholesterol Acyl Transferase Deficiency with Chronic Kidney Disease.
The Journal of the Association of Physicians of IndiaSequential kidney-liver transplantation from the same living donor for lecithin cholesterol acyl transferase deficiency.
Clinical transplantationApoA-IMilano phospholipid complex (ETC-216) infusion in human volunteers. Insights into the phenotypic characteristics of ApoA-IMilano carriers.
Pharmacological research[Gene-manipulated Adipocytes for the Treatment of Various Intractable Diseases].
Yakugaku zasshi : Journal of the Pharmaceutical Society of JapanFamilial lecithin:cholesterol acyltransferase deficiency: First-in-human treatment with enzyme replacement.
Journal of clinical lipidologyLipoprotein X Causes Renal Disease in LCAT Deficiency.
PloS oneAn unusual presentation of LCAT deficiency as nephrotic syndrome with normal serum HDL-C level.
Journal of nephropharmacologyCo-existence of classic familial lecithin-cholesterol acyl transferase deficiency and fish eye disease in the same family.
Indian journal of nephrologySafety and Tolerability of ACP-501, a Recombinant Human Lecithin:Cholesterol Acyltransferase, in a Phase 1 Single-Dose Escalation Study.
Circulation researchRole of LCAT in Atherosclerosis.
Journal of atherosclerosis and thrombosisHistiocytic and Nonhistiocytic Glomerular Lesions: Foam Cells and Their Mimickers.
American journal of kidney diseases : the official journal of the National Kidney FoundationLecithin:Cholesterol Acyltransferase (LCAT) Deficiency Promotes Differentiation of Satellite Cells to Brown Adipocytes in a Cholesterol-dependent Manner.
The Journal of biological chemistryBeta2-adrenergic activity modulates vascular tone regulation in lecithin:cholesterol acyltransferase knockout mice.
Vascular pharmacologyLack of LCAT reduces the LPS-neutralizing capacity of HDL and enhances LPS-induced inflammation in mice.
Biochimica et biophysica actaStructure and function of lysosomal phospholipase A2 and lecithin:cholesterol acyltransferase.
Nature communicationsFamilial lecithin-cholesterol acyltransferase (LCAT) deficiency; a differential of proteinuria.
Journal of nephropathologyA robust all-atom model for LCAT generated by homology modeling.
Journal of lipid researchAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Deficiência de LCAT.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Deficiência de LCAT
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Lecithin-cholesterol acyltransferase deficiency with the finding of "zebra bodies": A diagnostic challenge in the context of suspected Fabry disease.
- Lecithin-Cholesterol Acyltransferase Deficiency as a Rare Cause of Bilateral Corneal Opacities: A Case Report of a Novel Frameshift Mutation.
- A Case of Acquired LCAT Deficiency with the Discrepancy between Spontaneous Resolution of Proteinuria and Continually Low HDL Cholesterol Levels.
- Loss of LCAT function aggravates metabolic-associated steatohepatitis (MASH) in golden Syrian hamster.
- Quantitative Assessment of Visual Function in Japanese Patients With Lecithin-Cholesterol Acyltransferase Gene Abnormalities: A Case-Control Study.
- IgG3κ Monoclonal Membranous Nephropathy Associated With Acquired Lecithin Cholesterol Acyltransferase Deficiency.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:650(Orphanet)
- MONDO:0018999(MONDO)
- GARD:16539(GARD (NIH))
- Variantes catalogadas(ClinVar)
- Busca completa no PubMed(PubMed)
- Q56014242(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar