Introdução
O que você precisa saber de cara
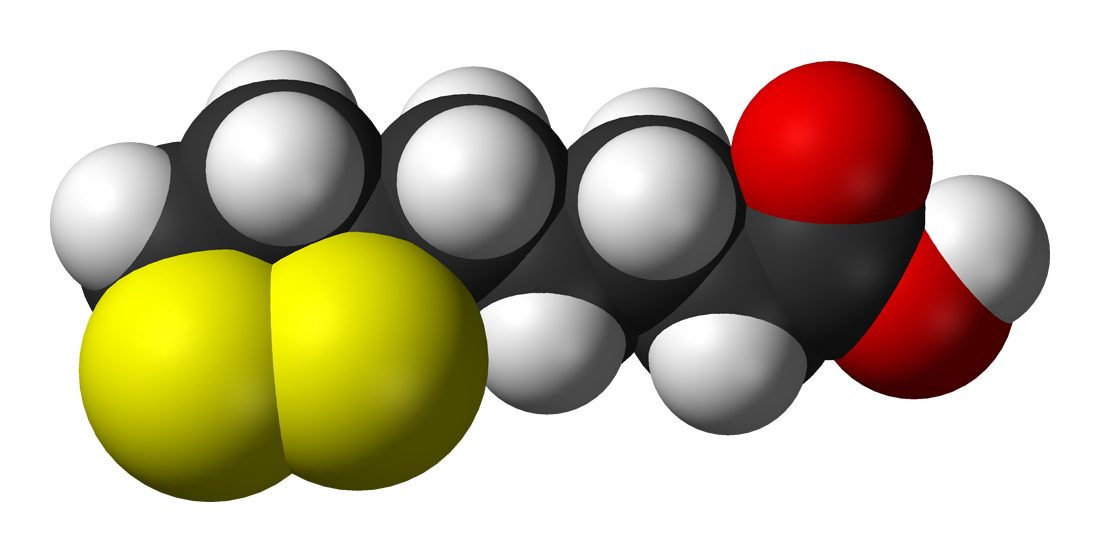
O ácido lipoico (AL), também conhecido como ácido α-lipoico, ácido alfa-lipoico (ALA) e ácido tióctico, é um composto organossulfurado derivado do ácido caprílico (ácido octanoico). O ALA, que é produzido normalmente em animais, é essencial para o metabolismo aeróbico. Também está disponível como suplemento dietético ou medicamento farmacêutico em alguns países. O lipoato é a base conjugada do ácido lipoico e a forma mais prevalente de AL sob condições fisiológicas. Apenas o enantiômero (R)-(+)-(RLA) existe na natureza. O RLA é um cofator essencial de muitos processos.
Escala de raridade
<1/50kMuito rara
1/20kRara
1/10kPouco freq.
1/5kIncomum
1/2k
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Partes do corpo afetadas
+ 13 sintomas em outras categorias
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 26 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Genes associados
1 gene identificado com associação a esta condição. Padrão de herança: Autosomal recessive.
Lipoyl amidotransferase that catalyzes the transfer of lipoyl moieties from lipoyl-protein H of the glycine cleavage system (lipoyl-GCSH) to E2 subunits of the pyruvate dehydrogenase complex (PDCE2) (PubMed:29987032). Unable to catalyze the transfer of octanoyl from octanoyl-GCSH to PDCE2 (PubMed:29987032). In vitro, it is also able to catalyze the transfer of the lipoyl group from lipoyl-AMP to the specific lysine residue of lipoyl domains of lipoate-dependent enzymes but this reaction may not
Mitochondrion
Lipoyltransferase 1 deficiency
An autosomal recessive disorder due to a defect in lipoic acid metabolism, resulting in severe lactic acidosis and metabolic decompensation. Variable clinical manifestations include delayed psychomotor development, severe hypotonia, dystonia, loss of head control, coma, bradycardia, and pulmonary hypertension.
Variantes genéticas (ClinVar)
39 variantes patogênicas registradas no ClinVar.
Classificação de variantes (ClinVar)
Distribuição de 17 variantes classificadas pelo ClinVar.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Déficit de lipoil transferase 1
Selecione um estado ou use sua localização para ver resultados.
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Pesquisa e ensaios clínicos
Nenhum ensaio clínico registrado para esta condição.
Publicações mais relevantes
Inhibition of DHHC9-mediated CD36 palmitoylation lessens high-fat diet (HFD)-induced impairment of pubertal mammary gland development through the JNK-ERK pathway.
CD36, a vital fatty acid translocase, has been reported to participate in multiple physiological functions through palmitoylation mediated by zinc finger Asp-His-His-Cys-type palmitoyltransferases (DHHCs). This study aimed to investigate the possible involvement of DHHC-mediated CD36 palmitoylation in high-fat diet (HFD)-induced impairment of pubertal mammary gland development and explore the underlying mechanisms involved. Palmitic acid (PA)-treated HC11 cells were used as the in vitro high-fat model, and the cell proliferation was examined by 5-Ethynyl-2'-deoxyuridine (EdU) incorporation assay. The palmitoylation of CD36 was determined by the acyl-biotin exchange (ABE) method. The expression of CD36, proliferative genes, and signaling molecules was detected by immunoblotting. The cellular localization of CD36 was determined by immunofluorescence. The bindings of CD36 with zinc finger DHHC-type palmitoyltransferases 9 (DHHC9) or Fyn/Lyn were detected by co-immunoprecipitation (Co-IP). The palmitoylation inhibitor 2-bromopalmitate (2BP), DHHC9 knockdown, and point mutation of CD36 cysteine residues were applied to construct a CD36 palmitoylation deficiency model in vitro to investigate the effects of CD36 palmitoylation on HC11 proliferation. In vivo, the pubertal mice were treated with HFD and/or 2BP. Mammary gland morphology was determined by whole mount staining, and the underlying mechanisms were verified by the methods used in the in vitro system. In vitro, the palmitoylation inhibitor 2BP eliminated PA-inhibited HC11 proliferation and inhibited CD36 palmitoylation and localization on the plasma membrane. Meanwhile, the binding of DHHC9 and CD36 in PA-treated HC11 cells was repressed by 2BP. In addition, both knockdown of DHHC9 and point mutation of CD36 cysteine residues suppressed the membrane palmitoylation and localization of CD36 and stimulated the proliferation of PA-treated HC11 cells. Furthermore, in PA-treated HC11 cells, the inhibition of CD36 palmitoylation, the knockdown of DHHC9, and the mutation of CD36 cysteine residues resulted in decreased formation of the CD36/Fyn/Lyn complex. Correspondingly, the downstream c-jun n-terminal kinase 1 (JNK1) pathway was inhibited, and the extracellular signal-regulated kinase 1/2 (ERK1/2) pathway was activated. Moreover, inhibition of the JNK pathway with SP600125 promoted the proliferation of PA-treated HC11 cells via activation of the ERK1/2 pathway. In vivo, the palmitoylation inhibitor 2BP ameliorated HFD-induced impairment of mammary gland development in pubertal female mice, which was associated with a decrease in DHHC9-mediated CD36 palmitoylation in the plasma membrane, a reduction in the CD36/Fyn/Lyn complex, inhibition of the JNK1 pathway, and activation of the ERK1/2 pathway. This study revealed that inhibition of DHHC9-mediated CD36 palmitoylation mitigated HFD-induced impairment of pubertal mammary gland development via the JNK1-ERK1/2 pathway.
zDHHC8 suppresses infection-induced necroptosis in monocyte/macrophages through palmitoylation of necrosome components in large yellow croaker (Larimichthys crocea).
Palmitoylation, a reversible post-translational modification, regulates a wide range of cellular processes by modulating protein localization, stability, and interactions. The zinc finger Asp-His-His-Cys (zDHHC) family of palmitoyltransferases is central to this process, yet their functions in immune cell death of lower vertebrates, remain poorly understood. Here, we investigate the role of zDHHC8 in modulating immune responses and necroptosis during infection in large yellow croaker (LczDHHC8). We demonstrate that LczDHHC8 is significantly upregulated in monocyte/macrophages (MO/MΦ) during Pseudomonas plecoglossicida infection and plays a key role in promoting pro-inflammatory cytokine production and M1 macrophage polarization. Through acyl-biotin exchange (ABE) assays and liquid chromatography-mass spectrometry (LC-MS/MS), we show that LczDHHC8 suppresses the palmitoylation of necrosome components, including receptor-interacting protein kinases 1 (RIPK1), receptor-interacting protein kinases 3 (RIPK3), and mixed lineage kinase domain-like protein (MLKL), thereby influencing necroptosis of MO/MΦ. LczDHHC8 deficiency exacerbates necroptosis, evidenced by increased phosphorylation of necroptosis-related proteins and enhanced cell death. Our findings suggest that LczDHHC8 may negatively regulates necroptosis through facilitating the palmitoylation of necrosome components, thereby influencing MO/MΦ function by modulating immune responses and cell survival during infection. This study provides novel insights into the immune regulatory roles of palmitoylation in teleosts and emphasizes the importance of LczDHHC8 in the modulation of necroptosis, which may have broader implications for understanding host-pathogen interactions and inflammatory responses in vertebrates.
Zdhhc1 deficiency mitigates foam cell formation and atherosclerosis by inhibiting PI3K-Akt-mTOR signaling pathway through facilitating the nuclear translocation of p110α.
Monocyte-to-macrophage differentiation and subsequent foam cell formation are key processes that contribute to plaque build-up during the progression of atherosclerotic lesions. Palmitoylation enzymes are known to play pivotal roles in the development and progression of inflammatory diseases. However, their specific impact on atherosclerosis development remains unclear. In this study, we discovered that the knockout of zDHHC1 in THP-1 cells, as well as Zdhhc1 in mice, markedly reduces the uptake of oxidized low-density lipoprotein (ox-LDL) by macrophages, thereby inhibiting foam cell formation. Moreover, the absence of Zdhhc1 in ApoE-/- mice significantly suppresses atherosclerotic plaque formation. Mass spectrometry coupled with bioinformatic analysis revealed an enrichment of the PI3K-Akt-mTOR signaling pathway. Consistent with this, we observed that knockout of zDHHC1 significantly decreases the palmitoylation levels of p110α, a crucial subunit of PI3K. Notably, the deletion of Zdhhc1 facilitates the nuclear translocation of p110α in macrophages, leading to a significant reduction in the downstream phosphorylation of Akt at Ser473 and mTOR at Ser2448. This cascade results in a decreased number of macrophages within plaques and ultimately mitigates the severity of atherosclerosis. These findings unveil a novel role for zDHHC1 in regulating foam cell formation and the progression of atherosclerosis, suggesting it as a promising target for clinical intervention in atherosclerosis therapy.
ROS-dependent S-palmitoylation activates cleaved and intact gasdermin D.
Gasdermin D (GSDMD) is the common effector for cytokine secretion and pyroptosis downstream of inflammasome activation and was previously shown to form large transmembrane pores after cleavage by inflammatory caspases to generate the GSDMD N-terminal domain (GSDMD-NT)1-10. Here we report that GSDMD Cys191 is S-palmitoylated and that palmitoylation is required for pore formation. S-palmitoylation, which does not affect GSDMD cleavage, is augmented by mitochondria-generated reactive oxygen species (ROS). Cleavage-deficient GSDMD (D275A) is also palmitoylated after inflammasome stimulation or treatment with ROS activators and causes pyroptosis, although less efficiently than palmitoylated GSDMD-NT. Palmitoylated, but not unpalmitoylated, full-length GSDMD induces liposome leakage and forms a pore similar in structure to GSDMD-NT pores shown by cryogenic electron microscopy. ZDHHC5 and ZDHHC9 are the major palmitoyltransferases that mediate GSDMD palmitoylation, and their expression is upregulated by inflammasome activation and ROS. The other human gasdermins are also palmitoylated at their N termini. These data challenge the concept that cleavage is the only trigger for GSDMD activation. They suggest that reversible palmitoylation is a checkpoint for pore formation by both GSDMD-NT and intact GSDMD that functions as a general switch for the activation of this pore-forming family.
Reduction of DHHC5-mediated beclin 1 S-palmitoylation underlies autophagy decline in aging.
Autophagy is a lysosome-dependent degradation pathway essential for cellular homeostasis, which decreases with age. However, it is unclear how aging induces autophagy decline. Here we show the role of protein S-palmitoylation in autophagy. We identify the palmitoyl acyltransferase DHHC5 as a regulator of autophagy by mediating the palmitoylation of beclin 1, which in turn promotes the formation of ATG14L-containing class III phosphatidylinositol-3-kinase complex I and its lipid kinase activity by promoting the hydrophobic interactions between beclin 1 and adapter proteins ATG14L and VPS15. In aging brains of human and nonhuman primate, the levels of DHHC5 exhibit a marked decrease in expression. We show that DHHC5 deficiency in neurons leads to reduced cellular protein homeostasis in two established murine models of Alzheimer's disease, which exaggerates neurodegeneration in an autophagy-dependent manner. These findings identify reduction of DHHC5-mediated beclin 1 S-palmitoylation as an underlying mechanism by which aging induces autophagy decline.
📚 EuropePMCmostrando 32
Inhibition of DHHC9-mediated CD36 palmitoylation lessens high-fat diet (HFD)-induced impairment of pubertal mammary gland development through the JNK-ERK pathway.
Cellular & molecular biology letterszDHHC8 suppresses infection-induced necroptosis in monocyte/macrophages through palmitoylation of necrosome components in large yellow croaker (Larimichthys crocea).
International journal of biological macromoleculesZdhhc1 deficiency mitigates foam cell formation and atherosclerosis by inhibiting PI3K-Akt-mTOR signaling pathway through facilitating the nuclear translocation of p110α.
Biochimica et biophysica acta. Molecular basis of diseaseEngineered bacterial lipoate protein ligase A (lplA) restores lipoylation in cell models of lipoylation deficiency.
The Journal of biological chemistryZDHHC7-mediated S-palmitoylation of ATG16L1 facilitates LC3 lipidation and autophagosome formation.
AutophagyCaveolin-2 palmitoylation turnover facilitates insulin receptor substrate-1-directed lipid metabolism by insulin receptor tyrosine kinase.
Biochimica et biophysica acta. Molecular basis of diseaseROS-dependent S-palmitoylation activates cleaved and intact gasdermin D.
NaturezDHHC20-driven S-palmitoylation of CD80 is required for its costimulatory function.
Acta pharmacologica SinicaReduction of DHHC5-mediated beclin 1 S-palmitoylation underlies autophagy decline in aging.
Nature structural & molecular biologyAlpha-lipoic acid supplementation corrects pathological alterations in cellular models of pantothenate kinase-associated neurodegeneration with residual PANK2 expression levels.
Orphanet journal of rare diseasesInvolvement of ZDHHC9 in lung adenocarcinoma: regulation of PD-L1 stability via palmitoylation.
In vitro cellular & developmental biology. AnimalDHHC21 deficiency attenuates renal dysfunction during septic injury.
Scientific reportsIn a mouse model of INCL reduced S-palmitoylation of cytosolic thioesterase APT1 contributes to microglia proliferation and neuroinflammation.
Journal of inherited metabolic diseaseStructure of the dihydrolipoamide succinyltransferase (E2) component of the human alpha-ketoglutarate dehydrogenase complex (hKGDHc) revealed by cryo-EM and cross-linking mass spectrometry: Implications for the overall hKGDHc structure.
Biochimica et biophysica acta. General subjectsGenetic dissection of the mitochondrial lipoylation pathway in yeast.
BMC biologyA STAT3 palmitoylation cycle promotes TH17 differentiation and colitis.
NatureCD36 facilitates fatty acid uptake by dynamic palmitoylation-regulated endocytosis.
Nature communicationsDynamic palmitoylation events following T-cell receptor signaling.
Communications biologyPalmitoyl Acyltransferase Activity of ZDHHC13 Regulates Skin Barrier Development Partly by Controlling PADi3 and TGM1 Protein Stability.
The Journal of investigative dermatologyFatty acids and cancer-amplified ZDHHC19 promote STAT3 activation through S-palmitoylation.
NatureFunctional Assessment of Lipoyltransferase-1 Deficiency in Cells, Mice, and Humans.
Cell reportsThe dual roles of RPE65 S-palmitoylation in membrane association and visual cycle function.
Scientific reportsReduction of mitochondrial 3-oxoacyl-ACP synthase (OXSM) by hyperglycemia is associated with deficiency of α-lipoic acid synthetic pathway in kidney of diabetic mice.
Biochemical and biophysical research communicationsPoldip2 is an oxygen-sensitive protein that controls PDH and αKGDH lipoylation and activation to support metabolic adaptation in hypoxia and cancer.
Proceedings of the National Academy of Sciences of the United States of AmericaZdhhc13-dependent Drp1 S-palmitoylation impacts brain bioenergetics, anxiety, coordination and motor skills.
Scientific reportsBiallelic Mutations in LIPT2 Cause a Mitochondrial Lipoylation Defect Associated with Severe Neonatal Encephalopathy.
American journal of human geneticsDiscovery and Characterization of the 3-Hydroxyacyl-ACP Dehydratase Component of the Plant Mitochondrial Fatty Acid Synthase System.
Plant physiologyPalmitoyl acyltransferase Aph2 in cardiac function and the development of cardiomyopathy.
Proceedings of the National Academy of Sciences of the United States of AmericaA phosphopantetheinyl transferase that is essential for mitochondrial fatty acid biosynthesis.
The Plant journal : for cell and molecular biologyEssential role of flotillin-1 palmitoylation in the intracellular localization and signaling function of IGF-1 receptor.
Journal of cell scienceELMOD2 is anchored to lipid droplets by palmitoylation and regulates adipocyte triglyceride lipase recruitment.
Molecular biology of the cellFatty acylated caveolin-2 is a substrate of insulin receptor tyrosine kinase for insulin receptor substrate-1-directed signaling activation.
Biochimica et biophysica actaAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Déficit de lipoil transferase 1.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Déficit de lipoil transferase 1
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Inhibition of DHHC9-mediated CD36 palmitoylation lessens high-fat diet (HFD)-induced impairment of pubertal mammary gland development through the JNK-ERK pathway.
- zDHHC8 suppresses infection-induced necroptosis in monocyte/macrophages through palmitoylation of necrosome components in large yellow croaker (Larimichthys crocea).
- Zdhhc1 deficiency mitigates foam cell formation and atherosclerosis by inhibiting PI3K-Akt-mTOR signaling pathway through facilitating the nuclear translocation of p110α.
- ROS-dependent S-palmitoylation activates cleaved and intact gasdermin D.
- Reduction of DHHC5-mediated beclin 1 S-palmitoylation underlies autophagy decline in aging.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:401862(Orphanet)
- OMIM OMIM:616299(OMIM)
- MONDO:0014576(MONDO)
- GARD:12680(GARD (NIH))
- Variantes catalogadas(ClinVar)
- Q55784889(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar