Introdução
O que você precisa saber de cara
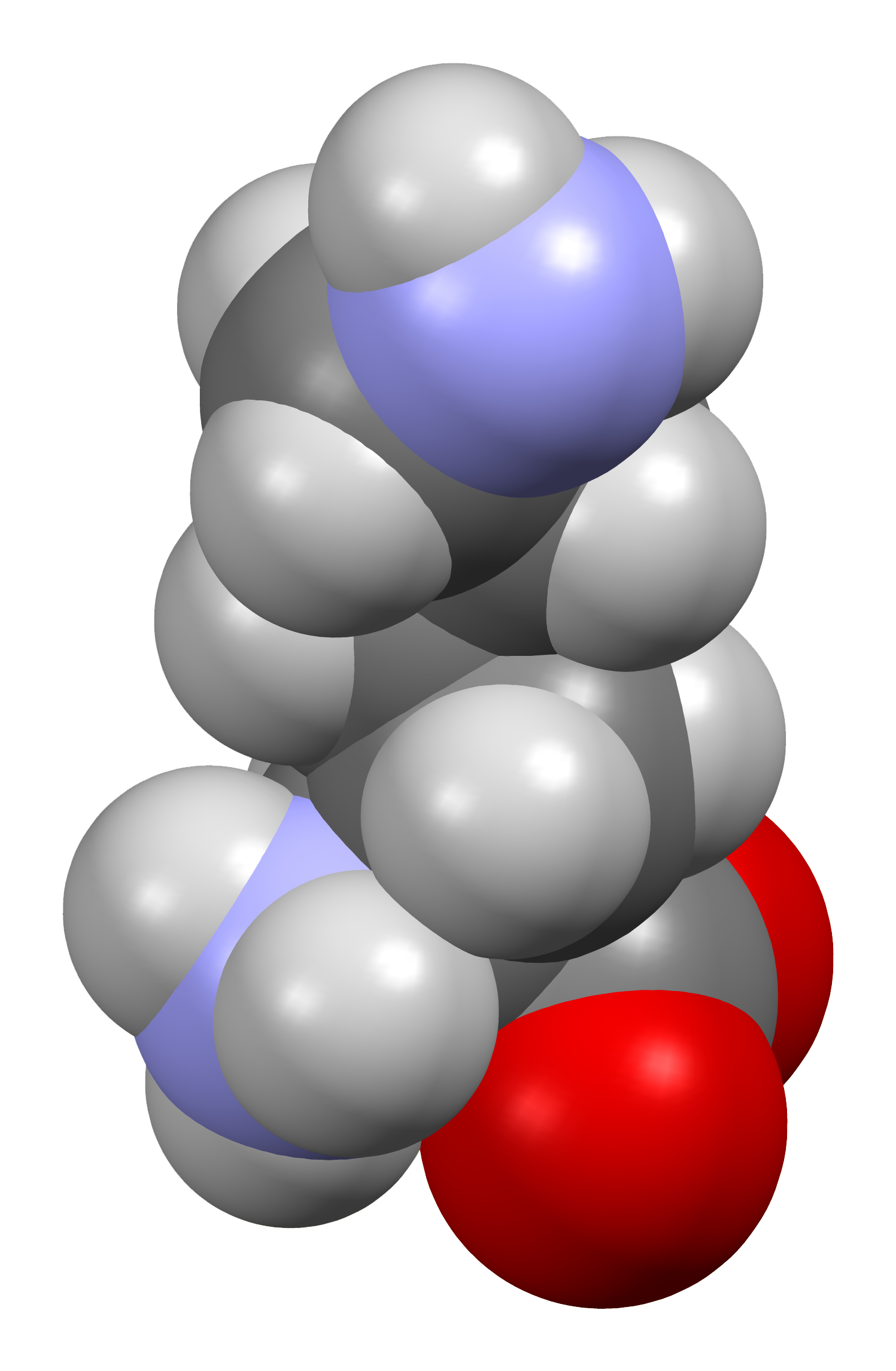
A lisina é um α-aminoácido que é um precursor de muitas proteínas. A lisina contém um grupo α-amino, um grupo α-ácido carboxílico e uma cadeia lateral (CH2)4NH2, sendo assim classificada como um aminoácido básico, carregado e alifático. É codificada pelos códons AAA e AAG. Como quase todos os outros aminoácidos, o carbono α é quiral e a lisina pode se referir a qualquer um dos enantiômeros ou a uma mistura racêmica de ambos. Para os propósitos deste artigo, a lisina se referirá ao enantiômero biologicamente ativo L-lisina, onde o carbono α está na configuração S.
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Partes do corpo afetadas
+ 42 sintomas em outras categorias
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 88 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Genes associados
2 genes identificados com associação a esta condição.
Bifunctional enzyme that catalyzes the first two steps in lysine degradation
Mitochondrion
Hyperlysinemia, 1
An autosomal recessive metabolic condition with variable clinical features. Some patients present with non-specific seizures, hypotonia, or mildly delayed psychomotor development, and increased serum lysine and pipecolic acid on laboratory analysis. However, about half of the probands are reported to be asymptomatic, and hyperlysinemia is generally considered to be a benign metabolic variant.
2-oxoadipate dehydrogenase (E1a) component of the 2-oxoadipate dehydrogenase complex (OADHC) (PubMed:29191460, PubMed:29752936, PubMed:32303640, PubMed:32633484, PubMed:32695416). Participates in the first step, rate limiting for the overall conversion of 2-oxoadipate (alpha-ketoadipate) to glutaryl-CoA and CO(2) catalyzed by the whole OADHC (PubMed:29191460, PubMed:32695416). Catalyzes the irreversible decarboxylation of 2-oxoadipate via the thiamine diphosphate (ThDP) cofactor and subsequent t
Mitochondrion
Charcot-Marie-Tooth disease, axonal, type 2Q
An axonal form of Charcot-Marie-Tooth disease, a disorder of the peripheral nervous system, characterized by progressive weakness and atrophy, initially of the peroneal muscles and later of the distal muscles of the arms. Charcot-Marie-Tooth disease is classified in two main groups on the basis of electrophysiologic properties and histopathology: primary peripheral demyelinating neuropathies (designated CMT1 when they are dominantly inherited) and primary peripheral axonal neuropathies (CMT2). Neuropathies of the CMT2 group are characterized by signs of axonal degeneration in the absence of obvious myelin alterations, normal or slightly reduced nerve conduction velocities, and progressive distal muscle weakness and atrophy.
Variantes genéticas (ClinVar)
186 variantes patogênicas registradas no ClinVar.
Classificação de variantes (ClinVar)
Distribuição de 1 variantes classificadas pelo ClinVar.
Vias biológicas (Reactome)
2 vias biológicas associadas aos genes desta condição.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Doença do metabolismo da lisina e hidroxilisina
Centros de Referência SUS
21 centros habilitados pelo SUS para Doença do metabolismo da lisina e hidroxilisina
Centros para Doença do metabolismo da lisina e hidroxilisina
Detalhes dos centros
Hospital Universitário Prof. Edgard Santos (HUPES)
R. Dr. Augusto Viana, s/n - Canela, Salvador - BA, 40110-060 · CNES 0003808
Serviço de Referência
Hospital de Apoio de Brasília (HAB)
AENW 3 Lote A Setor Noroeste - Plano Piloto, Brasília - DF, 70684-831 · CNES 0010456
Serviço de Referência
Hospital Estadual Infantil e Maternidade Alzir Bernardino Alves (HIABA)
Av. Min. Salgado Filho, 918 - Soteco, Vila Velha - ES, 29106-010 · CNES 6631207
Serviço de Referência
Hospital das Clínicas da UFG
Rua 235 QD. 68 Lote Área, Nº 285, s/nº - Setor Leste Universitário, Goiânia - GO, 74605-050 · CNES 2338424
Serviço de Referência
Hospital das Clínicas da UFMG
Av. Prof. Alfredo Balena, 110 - Santa Efigênia, Belo Horizonte - MG, 30130-100 · CNES 2280167
Serviço de Referência
NUPAD / Faculdade de Medicina UFMG
Av. Prof. Alfredo Balena, 189 - 5 andar - Centro, Belo Horizonte - MG, 30130-100 · CNES 2183226
Serviço de Referência
Hospital Universitário João de Barros Barreto
R. dos Mundurucus, 4487 - Guamá, Belém - PA, 66073-000 · CNES 2337878
Serviço de Referência
Hospital de Clínicas da Universidade Federal de Pernambuco
Av. Prof. Moraes Rego, 1235 - Cidade Universitária, Recife - PE, 50670-901 · CNES 2561492
Atenção Especializada
Instituto de Medicina Integral Prof. Fernando Figueira (IMIP)
R. dos Coelhos, 300 - Boa Vista, Recife - PE, 50070-902 · CNES 0000647
Serviço de Referência
Hospital de Clínicas da UFPR
R. Gen. Carneiro, 181 - Alto da Glória, Curitiba - PR, 80060-900 · CNES 2364980
Serviço de Referência
Hospital Universitário Pedro Ernesto (HUPE-UERJ)
Blvd. 28 de Setembro, 77 - Vila Isabel, Rio de Janeiro - RJ, 20551-030 · CNES 2280221
Serviço de Referência
Instituto Nacional de Saúde da Mulher, da Criança e do Adolescente Fernandes Figueira (IFF/Fiocruz)
Av. Rui Barbosa, 716 - Flamengo, Rio de Janeiro - RJ, 22250-020 · CNES 2269988
Serviço de Referência
Hospital Universitário Onofre Lopes (HUOL)
Av. Nilo Peçanha, 620 - Petrópolis, Natal - RN, 59012-300 · CNES 2408570
Atenção Especializada
Hospital São Lucas da PUCRS
Av. Ipiranga, 6690 - Jardim Botânico, Porto Alegre - RS, 90610-000 · CNES 2232928
Serviço de Referência
Hospital de Clínicas de Porto Alegre (HCPA)
Rua Ramiro Barcelos, 2350 Bloco A - Av. Protásio Alves, 211 - Bloco B e C - Santa Cecília, Porto Alegre - RS, 90035-903 · CNES 2237601
Serviço de Referência
Hospital Universitário da UFSC (HU-UFSC)
R. Profa. Maria Flora Pausewang - Trindade, Florianópolis - SC, 88036-800 · CNES 2560356
Serviço de Referência
Hospital das Clínicas da FMUSP
R. Dr. Ovídio Pires de Campos, 225 - Cerqueira César, São Paulo - SP, 05403-010 · CNES 2077485
Serviço de Referência
Hospital de Clínicas da UNICAMP
R. Vital Brasil, 251 - Cidade Universitária, Campinas - SP, 13083-888 · CNES 2748223
Serviço de Referência
Hospital de Clínicas de Ribeirão Preto (HCRP-USP)
R. Ten. Catão Roxo, 3900 - Vila Monte Alegre, Ribeirão Preto - SP, 14015-010 · CNES 2082187
Serviço de Referência
Instituto da Criança e do Adolescente (ICr-HCFMUSP)
Av. Dr. Enéas Carvalho de Aguiar, 647 - Cerqueira César, São Paulo - SP, 05403-000 · CNES 2081695
Serviço de Referência
UNIFESP / Hospital São Paulo
R. Napoleão de Barros, 715 - Vila Clementino, São Paulo - SP, 04024-002 · CNES 2688689
Serviço de Referência
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Pesquisa e ensaios clínicos
Nenhum ensaio clínico registrado para esta condição.
Publicações mais relevantes
Lactobacillus Johnsonii RS-7 Prevents Nrf2-Mediated Intestinal Oxidative Damage by Modulating Microbial Metabolites.
Oxidative stress is a crucial pathological basis for diverse intestinal diseases, such as inflammatory bowel disease (IBD). Previous studies show that Lactobacillus johnsonii RS-7 alleviates colitis in mice, but its effect on intestinal oxidative stress remains unclear. Considering this, the research aimed to explore the role and mechanism of this strain in alleviating oxidative damage in a colitis model. Colitis was induced in the mice using dextran sulfate sodium (DSS), and mice were subsequently randomly assigned to four groups. L. johnsonii RS-7 was evaluated for its effects on colon damage, oxidative stress, the Nrf2 pathway, and intestinal metabolites using histopathology, biochemical assays, qPCR, Western blotting, and non-targeted metabolomics. The results showed that L. johnsonii RS-7 significantly attenuated DSS-induced colonic tissue injury and reversed the DSS-associated decreases in serum CAT, T-SOD, GSH, and T-AOC levels (p < 0.05), along with an increase in malondialdehyde (MDA) (p < 0.05). Moreover, the strain significantly mitigated the down-regulation of antioxidant enzyme mRNA expression in colonic tissues (p < 0.05). Mechanistically, L. johnsonii RS-7 restored expression of Nrf2, NQO1, and HO-1 proteins in the colon and suppressed the up-regulation of Keap1. Non-targeted metabolomic analysis of intestinal contents further demonstrated that the strain significantly reduced levels of LPS 18:0 and triglycerides while concurrently increasing concentrations of DL-lysine and 5-hydroxylysine (p < 0.05). Correlation analysis indicated that LPS 18:0 and DL-lysine might be key metabolites regulating oxidative stress. The above results suggest that L. johnsonii RS-7 can alleviate colonic oxidative damage by regulating metabolites to activate the Nrf2 pathway, providing theoretical support for applying this strain to prevent intestinal diseases.
Diagnosis of glutaric aciduria type I based on neuroradiological findings: when neonatal screening fails.
Glutaric aciduria type I (GA-I) is an autosomal recessive disorder affecting the metabolism of lysine, hydroxylysine, and tryptophan. Patients present in the first age of life with an irreversible motor disorder, and neuroradiological imaging can suggest the presence of the condition. Biochemically, the disorder is characterized by elevated levels of glutaric and 3-hydroxy glutaric acid in the urine and glutarylcarnitine in the blood. This latter metabolite can be detected in dried blood spots, and the condition can therefore be included in some newborn screening programs. We present the case of a patient affected by GA-I that was undetected by newborn screening in whom the diagnosis was clinically oriented at the age of nine months by acute neurological symptoms, represented by persistent tonic seizures, and by neuroimaging showing bilateral signal alterations in the basal ganglia. Biochemical data, including glutarylcarnitine in dried blood spots and urinary excretion of glutaric acid, were normal in the acute phase and during follow-up. Molecular analysis confirmed a diagnosis of GA-I, showing a homozygous M405V variant of the GCDH gene, which is common in African populations and associated with a low-excretor phenotype characteristic of the disorder. In conclusion, although GA-I is included in neonatal screening programs, the biochemical markers in dried blood spots can be absent. Therefore, in patients of African origin, clinicians should maintain a high degree of vigilance in the presence of suggestive clinical and neuroradiological findings, even if biochemical parameters are normal.
Collagen IV biosynthesis: Intracellular choreography of post-translational modifications.
Collagen IV, an essential and evolutionarily conserved component of basement membranes, is one of the most extensively post-translationally modified proteins. Despite substantial research on fibrillar collagen biosynthesis, our understanding of collagen IV biosynthesis, including its post-translational modifications (PTMs), remains limited. Most PTMs occur intracellularly, primarily within the endoplasmic reticulum (ER). In this review, we examine the molecular ensemble that orchestrates collagen IV biosynthesis in the ER, highlighting the complex interplay between prolyl and lysyl hydroxylases, glycosyltransferases, and molecular chaperones. Furthermore, we discuss how defects in collagen IV and its PTMs contribute to various human pathologies, including Gould and Alport syndromes, fibrosis, and cancer. Understanding collagen IV PTMs is crucial for elucidating the molecular basis of these diseases and improving targeted treatments. By reviewing our knowledge of collagen IV biosynthesis, we illustrate how this evolutionarily conserved yet highly specialized molecular biosynthesis ensemble supports the diverse functions of collagen IV in health and disease.
Modeling Glutaric Aciduria Type I in human neuroblastoma cells recapitulates neuronal damage that can be rescued by gene replacement.
Glutaric Aciduria type I (GA1) is a rare neurometabolic disorder caused by mutations in the GDCH gene encoding for glutaryl-CoA dehydrogenase (GCDH) in the catabolic pathway of lysine, hydroxylysine and tryptophan. GCDH deficiency leads to increased concentrations of glutaric acid (GA) and 3-hydroxyglutaric acid (3-OHGA) in body fluids and tissues. These metabolites are the main triggers of brain damage. Mechanistic studies supporting neurotoxicity in mouse models have been conducted. However, the different vulnerability to some stressors between mouse and human brain cells reveals the need to have a reliable human neuronal model to study GA1 pathogenesis. In the present work we generated a GCDH knockout (KO) in the human neuroblastoma cell line SH-SY5Y by CRISPR/Cas9 technology. SH-SY5Y-GCDH KO cells accumulate GA, 3-OHGA, and glutarylcarnitine when exposed to lysine overload. GA or lysine treatment triggered neuronal damage in GCDH deficient cells. SH-SY5Y-GCDH KO cells also displayed features of GA1 pathogenesis such as increased oxidative stress vulnerability. Restoration of the GCDH activity by gene replacement rescued neuronal alterations. Thus, our findings provide a human neuronal cellular model of GA1 to study this disease and show the potential of gene therapy to rescue GCDH deficiency.
Glutaric Aciduria Presenting With an Acute Encephalitic Crisis: A Case Report.
Glutaric aciduria type 1 (GA1) is an organic aciduria inherited in an autosomal recessive pattern, with an occurrence rate of one in 100,000. It is caused by a deficiency of the enzyme glutaryl-CoA dehydrogenase (GCDH), encoded by the GCDH gene on chromosome 19. It is an important enzyme in the catabolism of amino acids such as tryptophan, lysine, and hydroxylysine. Its deficiency leads to the accumulation of organic acids such as glutaric acid and 3-hydroxyglutaric acid, which interfere with cerebral energy metabolism and cause neurological symptoms. Here, we discuss the case of a six-month-old male child who presented with status epilepticus following an eight-day history of fever. The child was started on anti-epileptics. Initially, the child was on non-invasive ventilation and was later intubated and taken on a mechanical ventilator. A magnetic resonance imaging (MRI) scan of the brain was performed, and the findings suggested GA1. The child was started on carnitine after samples were sent for tandem mass spectrometry (TMS) and urine gas chromatography-mass spectrometry (GC/MS), which came out to be positive for GA1. Despite the timely intervention, the child did not survive. Most cases exhibit movement disorders, with many presenting in acute encephalitic crises. Additionally, a significant portion of patients experience an insidious onset of the disease. An MRI of the brain shows widened Sylvian fissures in the majority of cases. Treatment of GA1 includes dietary modifications, including a low-lysine diet and administering carnitine. Early diagnosis and management result in decreased mortality and morbidity, which underscores the need for newborn screening.
Publicações recentes
A basic study for the molecular imaging of dual-energy CT in diagnosing anterior cruciate ligament injury of knee joint.
Intervertebral disc degeneration in warmblood horses: Histological and biochemical characterization.
Neutrophil Adhesion and the Release of the Free Amino Acid Hydroxylysine.
Free urinary glycosylated hydroxylysine as an indicator of altered collagen degradation in the mucopolysaccharidoses.
Glycation of type I collagen selectively targets the same helical domain lysine sites as lysyl oxidase-mediated cross-linking.
📚 EuropePMCmostrando 52
Lactobacillus Johnsonii RS-7 Prevents Nrf2-Mediated Intestinal Oxidative Damage by Modulating Microbial Metabolites.
Journal of food scienceCollagen IV biosynthesis: Intracellular choreography of post-translational modifications.
Matrix biology : journal of the International Society for Matrix BiologyDiagnosis of glutaric aciduria type I based on neuroradiological findings: when neonatal screening fails.
Italian journal of pediatricsGlutaric Aciduria Presenting With an Acute Encephalitic Crisis: A Case Report.
CureusLoss of the long form of Plod2 phenocopies contractures of Bruck syndrome-osteogenesis imperfecta.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchEffect of Dexamethasone on Adhesion of Human Neutrophils and Concomitant Secretion.
Biochemistry. BiokhimiiaModeling Glutaric Aciduria Type I in human neuroblastoma cells recapitulates neuronal damage that can be rescued by gene replacement.
Gene therapyAmino Acids Profile in Children with Acute Brucellosis.
Current pediatric reviewsIncreased susceptibility to quinolinic acid-induced seizures and long-term changes in brain oscillations in an animal model of glutaric acidemia type I.
Journal of neuroscience researchNew mechanistic insights to PLOD1-mediated human vascular disease.
Translational research : the journal of laboratory and clinical medicineNeutrophil Adhesion and the Release of the Free Amino Acid Hydroxylysine.
CellsLow excretor glutaric aciduria type 1 of insidious onset with dystonia and atypical clinical features, a diagnostic dilemma.
JIMD reports[Expert consensus for the diagnosis and treatment of glutaricacidemia type 1].
Zhonghua yi xue yi chuan xue za zhi = Zhonghua yixue yichuanxue zazhi = Chinese journal of medical geneticsTumour-associated macrophages drive stromal cell-dependent collagen crosslinking and stiffening to promote breast cancer aggression.
Nature materialsAmino acids serve as an important energy source for adult flukes of Clonorchis sinensis.
PLoS neglected tropical diseasesAdult-onset glutaric aciduria type I: rare presentation of a treatable disorder.
NeurogeneticsStructure-function analyses of the G729R 2-oxoadipate dehydrogenase genetic variant associated with a disorder of l-lysine metabolism.
The Journal of biological chemistryImpaired Gastric Hormone Regulation of Osteoblasts and Lysyl Oxidase Drives Bone Disease in Diabetes Mellitus.
JBMR plusPotential complementation effects of two disease-associated mutations in tetrameric glutaryl-CoA dehydrogenase is due to inter subunit stability-activity counterbalance.
Biochimica et biophysica acta. Proteins and proteomicsFree urinary glycosylated hydroxylysine as an indicator of altered collagen degradation in the mucopolysaccharidoses.
Journal of inherited metabolic diseasePathogenesis of brain damage in glutaric acidemia type I: Lessons from the genetic mice model.
International journal of developmental neuroscience : the official journal of the International Society for Developmental NeuroscienceMetabolomics reveals elevated urinary excretion of collagen degradation and epithelial cell turnover products in irritable bowel syndrome patients.
Metabolomics : Official journal of the Metabolomic SocietyHuman 2-Oxoglutarate Dehydrogenase and 2-Oxoadipate Dehydrogenase Both Generate Superoxide/H2O2 in a Side Reaction and Each Could Contribute to Oxidative Stress in Mitochondria.
Neurochemical researchThe lysyl oxidase like 2/3 enzymatic inhibitor, PXS-5153A, reduces crosslinks and ameliorates fibrosis.
Journal of cellular and molecular medicineMutations in PLOD3, encoding lysyl hydroxylase 3, cause a complex connective tissue disorder including recessive dystrophic epidermolysis bullosa-like blistering phenotype with abnormal anchoring fibrils and type VII collagen deficiency.
Matrix biology : journal of the International Society for Matrix BiologyNeutrophils as a source of branched-chain, aromatic and positively charged free amino acids.
Cell adhesion & migrationSwitching obese mothers to a healthy diet improves fetal hypoxemia, hepatic metabolites, and lipotoxicity in non-human primates.
Molecular metabolismGlycation of type I collagen selectively targets the same helical domain lysine sites as lysyl oxidase-mediated cross-linking.
The Journal of biological chemistryLong Lasting High Lysine Diet Aggravates White Matter Injury in Glutaryl-CoA Dehydrogenase Deficient (Gcdh-/-) Mice.
Molecular neurobiologyFabrication of nanofibrous microcarriers mimicking extracellular matrix for functional microtissue formation and cartilage regeneration.
BiomaterialsTwo Uneventful Pregnancies in a Woman with Glutaric Aciduria Type 1.
JIMD reportsEarly Diagnosed and Treated Glutaric Acidemia Type 1 Female Presenting with Subependymal Nodules in Adulthood.
JIMD reportsHydroxyhomocitrulline Is a Collagen-Specific Carbamylation Mark that Affects Cross-link Formation.
Cell chemical biologyCyclophilin B Deficiency Causes Abnormal Dentin Collagen Matrix.
Journal of proteome researchDistinct post-translational features of type I collagen are conserved in mouse and human periodontal ligament.
Journal of periodontal researchIdentification of 4-(Aminomethyl)-6-(trifluoromethyl)-2-(phenoxy)pyridine Derivatives as Potent, Selective, and Orally Efficacious Inhibitors of the Copper-Dependent Amine Oxidase, Lysyl Oxidase-Like 2 (LOXL2).
Journal of medicinal chemistryGlutaric Acidemia Type 1: A Case of Infantile Stroke.
JIMD reportsFKBP65-dependent peptidyl-prolyl isomerase activity potentiates the lysyl hydroxylase 2-driven collagen cross-link switch.
Scientific reportsGAI - distinct genotype and phenotype characteristics in reported Slovak patients.
Bratislavske lekarske listyMolecular insights into prolyl and lysyl hydroxylation of fibrillar collagens in health and disease.
Critical reviews in biochemistry and molecular biologyImmunolocalization of glutaryl-CoA dehydrogenase (GCDH) in adult and embryonic rat brain and peripheral tissues.
NeuroscienceProposed recommendations for diagnosing and managing individuals with glutaric aciduria type I: second revision.
Journal of inherited metabolic diseaseMolecular characterization of glycation-associated skin ageing: an alternative skin model to study in vitro antiglycation activity of topical cosmeceutical and pharmaceutical formulations.
The British journal of dermatologyDisentangling mechanisms involved in collagen pyridinoline cross-linking: The immunophilin FKBP65 is critical for dimerization of lysyl hydroxylase 2.
Proceedings of the National Academy of Sciences of the United States of AmericaPossible relationship between amino acids, aggression and psychopathy.
International journal of psychiatry in clinical practiceThe Extracellular Matrix Signature in Vein Graft Disease.
The Canadian journal of cardiologyHydPred: a novel method for the identification of protein hydroxylation sites that reveals new insights into human inherited disease.
Molecular bioSystemsClinical and molecular investigation in Chinese patients with glutaric aciduria type I.
Clinica chimica acta; international journal of clinical chemistryComprehensive Characterization of Glycosylation and Hydroxylation of Basement Membrane Collagen IV by High-Resolution Mass Spectrometry.
Journal of proteome researchNew Cases of DHTKD1 Mutations in Patients with 2-Ketoadipic Aciduria.
JIMD reportsSpectrum of mutations in Glutaryl-CoA dehydrogenase gene in glutaric aciduria type I--Study from South India.
Brain & developmentMultifactorial modulation of susceptibility to l-lysine in an animal model of glutaric aciduria type I.
Biochimica et biophysica actaAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Doença do metabolismo da lisina e hidroxilisina.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Doença do metabolismo da lisina e hidroxilisina
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Lactobacillus Johnsonii RS-7 Prevents Nrf2-Mediated Intestinal Oxidative Damage by Modulating Microbial Metabolites.
- Diagnosis of glutaric aciduria type I based on neuroradiological findings: when neonatal screening fails.
- Collagen IV biosynthesis: Intracellular choreography of post-translational modifications.Matrix biology : journal of the International Society for Matrix Biology· 2025· PMID 40618934mais citado
- Modeling Glutaric Aciduria Type I in human neuroblastoma cells recapitulates neuronal damage that can be rescued by gene replacement.
- Glutaric Aciduria Presenting With an Acute Encephalitic Crisis: A Case Report.
- A basic study for the molecular imaging of dual-energy CT in diagnosing anterior cruciate ligament injury of knee joint.
- Intervertebral disc degeneration in warmblood horses: Histological and biochemical characterization.
- Neutrophil Adhesion and the Release of the Free Amino Acid Hydroxylysine.
- Free urinary glycosylated hydroxylysine as an indicator of altered collagen degradation in the mucopolysaccharidoses.
- Glycation of type I collagen selectively targets the same helical domain lysine sites as lysyl oxidase-mediated cross-linking.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:289832(Orphanet)
- MONDO:0017351(MONDO)
- GARD:21155(GARD (NIH))
- Variantes catalogadas(ClinVar)
- Busca completa no PubMed(PubMed)
- Q55787002(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar