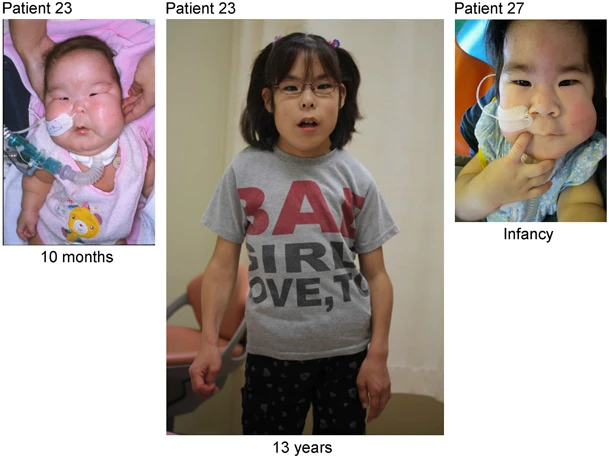
A síndrome de duplicação 14q32 é uma síndrome de anomalia cromossômica rara resultante da duplicação parcial do braço longo do cromossomo 14 que resulta em uma predisposição a uma série de neoplasias mieloproliferativas de início na idade adulta, incluindo leucemia mieloide aguda, leucemia mielomonocítica crônica e neoplasias mieloproliferativas, especialmente trombocitemia essencial. Pode ser observada progressão para mielofibrose e leucemia mieloide aguda secundária.
Introdução
O que você precisa saber de cara
A síndrome de duplicação 14q32 é uma síndrome de anomalia cromossômica rara resultante da duplicação parcial do braço longo do cromossomo 14 que resulta em uma predisposição a uma série de neoplasias mieloproliferativas de início na idade adulta, incluindo leucemia mieloide aguda, leucemia mielomonocítica crônica e neoplasias mieloproliferativas, especialmente trombocitemia essencial. Pode ser observada progressão para mielofibrose e leucemia mieloide aguda secundária.
Escala de raridade
<1/50kMuito rara
1/20kRara
1/10kPouco freq.
1/5kIncomum
1/2k
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 6 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Nenhum gene associado encontrado
Os dados genéticos desta condição ainda estão sendo catalogados.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Síndrome de duplicação 14q32
Selecione um estado ou use sua localização para ver resultados.
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Pesquisa e ensaios clínicos
Nenhum ensaio clínico registrado para esta condição.
Publicações mais relevantes
Rare structural variants, aneuploidies, and mosaicism in individuals with Mullerian aplasia detected by optical genome mapping.
The molecular basis of Mayer-Rokitansky-Kuster-Hauser (MRKH) syndrome remains largely unknown. Pathogenic variants in WNT4 and HNF1B have been confirmed in a small percent of individuals. A variety of copy number variants have been reported, but causal gene(s) remain to be identified. We hypothesized that rare structural variants (SVs) would be present in some individuals with MRKH, which could explain the genetic basis of the syndrome. Large molecular weight DNA was extracted from lymphoblastoid cells from 87 individuals with MRKH and available parents. Optical genome mapping (OGM) was performed to identify SVs, which were confirmed by another method (quantitative PCR, chromosomal microarray, karyotype, or fluorescent in situ hybridization) when possible. Thirty-four SVs that overlapped coding regions of genes with potential involvement in MRKH were identified, 14 of which were confirmed by a second method. These 14 SVs were present in 17/87 (19.5%) of probands with MRKH and included seven deletions, three duplications, one new translocation in 5/50 cells-t(7;14)(q32;q32), confirmation of a previously identified translocation-t(3;16)(p22.3;p13.3), and two aneuploidies. Of interest, three cases of mosaicism (3.4% of probands) were identified-25% mosaicism for trisomy 12, 45,X(75%)/46,XX (25%), and 10% mosaicism for a 7;14 translocation. Our study constitutes the first systematic investigation of SVs by OGM in individuals with MRKH. We propose that OGM is a promising method that enables a comprehensive investigation of a variety of SVs in a single assay including cryptic translocations and mosaic aneuploidies. These observations suggest that mosaicism could play a role in the genesis of MRKH.
Microdeletions in 1q21 and 8q12.1 depict two additional molecular subgroups of Silver-Russell syndrome like phenotypes.
Silver-Russell syndrome (SRS) is a genetic disorder characterized by intrauterine and postnatal growth restriction, relative macrocephaly at birth, body asymmetry and typical facial features. Clinical and molecular heterogeneity is described in SRS. Common causes are loss of methylation of the imprinting center 1 in 11p15 and maternal uniparental disomy of chromosome 7. Other genetic alterations include disturbances of imprinted regions in 14q32, 7q32 and 11p15 as well as submicroscopic deletions and duplications. Single nucleotide variants in genes like IGF2, HMGA2, PLAG1, CDKN1C have also been identified in patients with SRS phenotypes. However, routine molecular diagnostics usually focus on 11p15 and chromosome 7, while less frequent causes are not systematically addressed. Here we report two patients with SRS features in which molecular karyotyping revealed microdeletions in 1q21 and 8q12.1 respectively. In a 3.5-year-old girl with postnatal growth restriction, feeding difficulties, relative macrocephaly and distinct SRS features a 2 Mb deletion in 1q21.1q21.2 was identified. Our second case is a 1.5-year-old boy with intrauterine and postnatal growth restriction, feeding difficulties and distinct facial features with a 77 kb deletion in 8q12.1 affecting PLAG1 as the only protein-encoding gene with known function. The 1q21 region has not yet been assigned as an SRS region, although six patients with the same deletion and SRS features including relative macrocephaly have been described before. This new case adds to the evidence that distal 1q21 should be annotated as an SRS candidate region. The PLAGL1 alteration is the smallest deletion in 8q12.1 ever reported in a patient with SRS phenotype and it finally confirms that PLAG1 is the SRS causing gene in 8q12.1. To increase the diagnostic yield in patients with suspected SRS, we recommend both molecular karyotyping and next generation sequencing-based approaches.
Germline ATG2B/GSKIP-containing 14q32 duplication predisposes to early clonal hematopoiesis leading to myeloid neoplasms.
The germline predisposition associated with the autosomal dominant inheritance of the 14q32 duplication implicating ATG2B/GSKIP genes is characterized by a wide clinical spectrum of myeloid neoplasms. We analyzed 12 asymptomatic carriers and 52 patients aged 18-74 years from six families, by targeted sequencing of 41 genes commonly mutated in myeloid malignancies. We found that 75% of healthy carriers displayed early clonal hematopoiesis mainly driven by TET2 mutations. Molecular landscapes of patients revealed two distinct routes of clonal expansion and leukemogenesis. The first route is characterized by the clonal dominance of myeloproliferative neoplasms (MPN)-driver events associated with TET2 mutations in half of cases and mutations affecting splicing and/or the RAS pathway in one-third of cases, leading to the early development of MPN, mostly essential thrombocythemia, with a high risk of transformation (50% after 10 years). The second route is distinguished by the absence of MPN-driver mutations and leads to AML without prior MPN. These patients mostly harbored a genomic landscape specific to acute myeloid leukemia secondary to myelodysplastic syndrome. An unexpected result was the total absence of DNMT3A mutations in this cohort. Our results suggest that the germline duplication constitutively mimics hematopoiesis aging by favoring TET2 clonal hematopoiesis.
New euchromatic variant dup(11)(p15.3p15.1) transmitted through two generations defined by low coverage whole genome sequencing.
We report on a 14-year old boy, his father, and his paternal uncle, all three carriers of a duplication of chromosomal region 11p15.3-p15.1. The aberration was transmitted by the grandmother, who is carrier of a balanced insertion 46,XX,ins(14;11)(q32.1;p15.3p15.1). In order to determine the precise molecular basis of this structural variant, we performed low-coverage whole genome sequencing on the boy's father. This approach allowed precise determination of the genomic breakpoints and revealed a duplication of 6.9 Mb, centromeric to the Beckwith-Wiedemann/Silver-Russell syndrome critical region in 11p15.5, that inserted in inverse orientation into 14q32.12 (according to HGVS nomenclature: NC_000014.8:g.92871000_92871001ins[NC_000011.9:g.12250642_19165928inv;T]). To our knowledge, this is the first report of a duplication of 11p15.3-p15.1 involving more than 40 genes and transmitted through two generations without apparent clinical effects.
Insights from the genetic characterization of central precocious puberty associated with multiple anomalies.
Is there an (epi)genetic basis in patients with central precocious puberty (CPP) associated with multiple anomalies that unmasks underlying mechanisms or reveals novel genetic findings related to human pubertal control? In a group of 36 patients with CPP associated with multiple phenotypes, pathogenic or likely pathogenic (epi)genetic defects were identified in 12 (33%) patients, providing insights into the genetics of human pubertal control. A few studies have described patients with CPP associated with multiple anomalies, but without making inferences on causalities of CPP. Genetic-molecular studies of syndromic cases may reveal disease genes or mechanisms, as the presentation of such patients likely indicates a genetic disorder. This translational study was based on a genetic-molecular analysis, including genome-wide high throughput methodologies, for searching structural or sequence variants implicated in CPP and DNA methylation analysis of candidate regions. A cohort of 197 patients (188 girls) with CPP without structural brain lesions was submitted to a detailed clinical evaluation, allowing the selection of 36 unrelated patients (32 girls) with CPP associated with multiple anomalies. Pathogenic allelic variants of genes known to cause monogenic CPP (KISS1R, KISS1, MKRN3 and DLK1) had been excluded in the entire cohort (197 patients). All selected patients with CPP associated with multiple anomalies (n = 36) underwent methylation analysis of candidate regions and chromosomal microarray analysis. A subset (n = 9) underwent whole-exome sequencing, due to presenting familial CPP and/or severe congenital malformations and neurocognitive abnormalities. Among the 36 selected patients with CPP, the more prevalent associated anomalies were metabolic, growth and neurocognitive conditions. In 12 (33%) of them, rare genetic abnormalities were identified: six patients presented genetic defects in loci known to be involved with CPP (14q32.2 and 7q11.23), whereas the other six presented defects in candidate genes or regions. In detail, three patients presented hypomethylation of DLK1/MEG3:IG-DMR (14q32.2 disruption or Temple syndrome), resulting from epimutation (n = 1) or maternal uniparental disomy of chromosome 14 (n = 2). Seven patients presented pathogenic copy number variants: three with de novo 7q11.23 deletions (Williams-Beuren syndrome), three with inherited Xp22.33 deletions, and one with de novo 1p31.3 duplication. Exome sequencing revealed potential pathogenic variants in two patients: a sporadic female case with frameshift variants in TNRC6B and AREL1 and a familial male case with a missense substitution in UGT2B4 and a frameshift deletion in MKKS. The selection of patients was based on a retrospective clinical characterization, lacking a longitudinal inclusion of consecutive patients. In addition, future studies are needed, showing the long-term (mainly reproductive) outcomes in the included patients, as most of them are not in adult life yet. The results highlighted the relevance of an integrative clinical-genetic approach in the elucidation of mechanisms and factors involved in pubertal control. Chromosome 14q32.2 disruption indicated the loss of imprinting of DLK1 as a probable mechanism of CPP. Two other chromosomal regions (7q11.23 and Xp22.33) represented new candidate loci potentially involved in this disorder of pubertal timing. This work was supported by grant number 2018/03198-0 (to A.P.M.C.) and grant number 2013/08028-1 (to A.C.V.K) from the São Paulo Research Foundation (FAPESP), and grant number 403525/2016-0 (to A.C.L.) and grant number 302849/2015-7 (to A.C.L.) and grant number 141625/2016-3 (to A.C.V.K) from the National Council for Scientific and Technological Development (CNPq). The authors have nothing to disclose. N/A.
Publicações recentes
Ver todas no PubMed📚 EuropePMCmostrando 15
Rare structural variants, aneuploidies, and mosaicism in individuals with Mullerian aplasia detected by optical genome mapping.
Human geneticsMicrodeletions in 1q21 and 8q12.1 depict two additional molecular subgroups of Silver-Russell syndrome like phenotypes.
Molecular cytogeneticsGermline ATG2B/GSKIP-containing 14q32 duplication predisposes to early clonal hematopoiesis leading to myeloid neoplasms.
LeukemiaNew euchromatic variant dup(11)(p15.3p15.1) transmitted through two generations defined by low coverage whole genome sequencing.
American journal of medical genetics. Part AInsights from the genetic characterization of central precocious puberty associated with multiple anomalies.
Human reproduction (Oxford, England)Prenatal diagnosis and molecular cytogenetic characterization of a small supernumerary marker chromosome (sSMC) inherited from her mosaic sSMC(15) mother and a literature review.
Taiwanese journal of obstetrics & gynecologyProfiling allele-specific gene expression in brains from individuals with autism spectrum disorder reveals preferential minor allele usage.
Nature neuroscienceSingle-nucleotide polymorphism-based chromosomal microarray analysis provides clues and insights into disease mechanisms.
Ultrasound in obstetrics & gynecology : the official journal of the International Society of Ultrasound in Obstetrics and GynecologyGermline duplication of ATG2B and GSKIP genes is not required for the familial myeloid malignancy syndrome associated with the duplication of chromosome 14q32.
LeukemiaMajor Contribution of Genomic Copy Number Variation in Syndromic Congenital Heart Disease: The Use of MLPA as the First Genetic Test.
Molecular syndromologyPrenatal diagnosis and molecular cytogenetic characterization of a de novo unbalanced reciprocal translocation of der(9)t(9;14)(p24.2;q32.11) associated with 9p terminal deletion and 14q distal duplication.
Taiwanese journal of obstetrics & gynecologyNSD1 duplication in Silver-Russell syndrome (SRS): molecular karyotyping in patients with SRS features.
Clinical geneticsA Familial 14q32.32q32.33 Duplication/17p13.3 Deletion Syndrome with Facial Anomalies and Moderate Intellectual Disability.
Cytogenetic and genome researchRare double-hit with two translocations involving IGH both, with BCL2 and BCL3, in a monoclonal B-cell lymphoma/leukemia.
Molecular cytogeneticsClinical features associated with copy number variations of the 14q32 imprinted gene cluster.
American journal of medical genetics. Part AAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Síndrome de duplicação 14q32.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Síndrome de duplicação 14q32
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Rare structural variants, aneuploidies, and mosaicism in individuals with Mullerian aplasia detected by optical genome mapping.
- Microdeletions in 1q21 and 8q12.1 depict two additional molecular subgroups of Silver-Russell syndrome like phenotypes.
- Germline ATG2B/GSKIP-containing 14q32 duplication predisposes to early clonal hematopoiesis leading to myeloid neoplasms.
- New euchromatic variant dup(11)(p15.3p15.1) transmitted through two generations defined by low coverage whole genome sequencing.
- Insights from the genetic characterization of central precocious puberty associated with multiple anomalies.
- A Recurrent De Novo Terminal Duplication of 14q32 in Korean Siblings Associated with Developmental Delay and Intellectual Disability, Growth Retardation, Facial Dysmorphism, and Cerebral Infarction: A Case Report and Literature Review.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:488280(Orphanet)
- OMIM OMIM:616604(OMIM)
- MONDO:0014707(MONDO)
- GARD:17890(GARD (NIH))
- Busca completa no PubMed(PubMed)
- Q55784945(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar