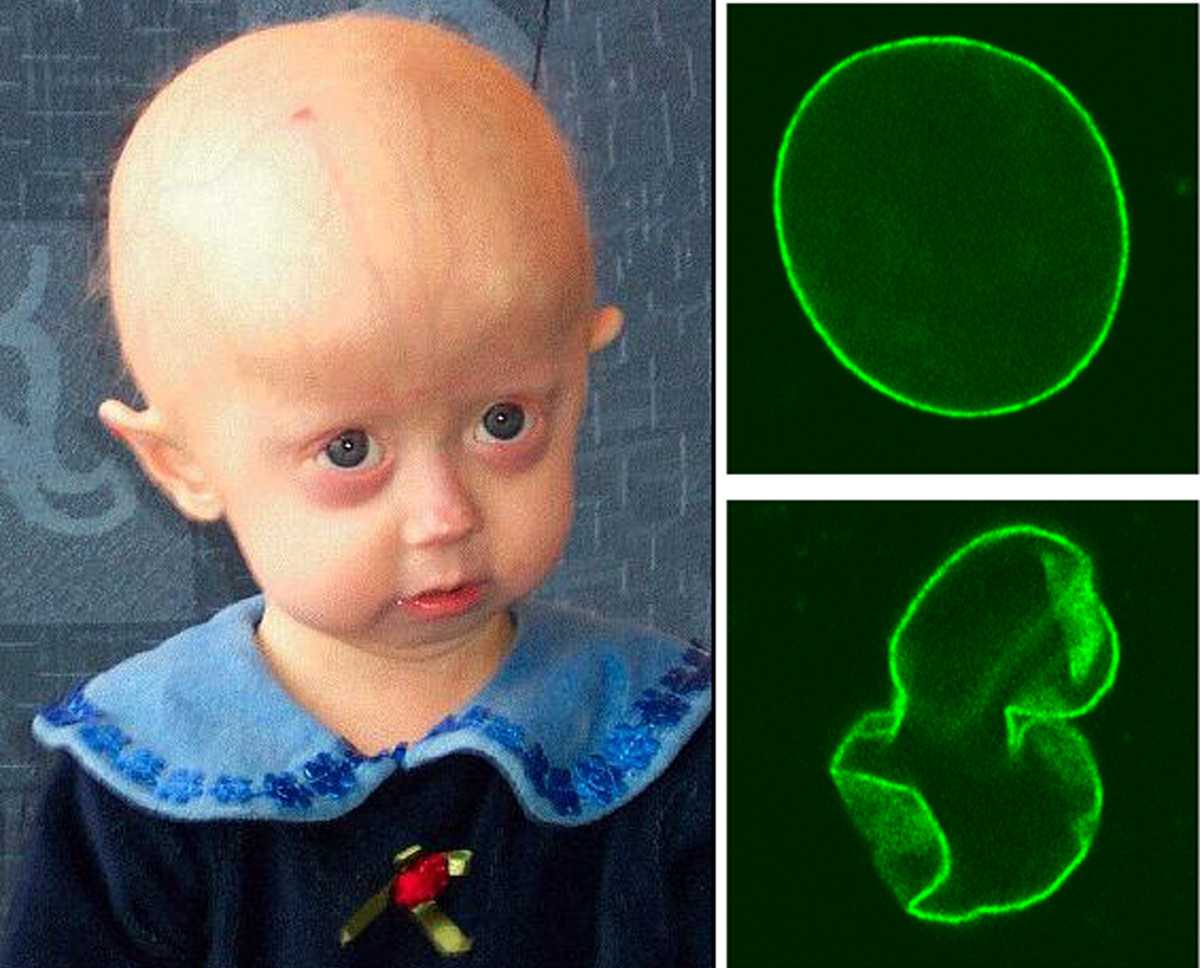
A síndrome de progéria de Hutchinson-Gilford é uma doença rara, fatal, autossômica dominante e de envelhecimento prematuro, que começa na infância e é caracterizada por redução do crescimento, retardo de crescimento, aparência facial típica (testa proeminente, olhos protuberantes, nariz fino com ponta em bico, lábios finos, micrognatia e orelhas salientes) e características dermatológicas distintas (alopecia generalizada, pele com aparência envelhecida, pele esclerótica e com covinhas no abdômen e extremidades, pele proeminente vasculatura, despigmentação, hipoplasia ungueal e perda de gordura subcutânea).
Introdução
O que você precisa saber de cara
A síndrome de progéria de Hutchinson-Gilford é uma doença rara, fatal, autossômica dominante e de envelhecimento prematuro, que começa na infância e é caracterizada por redução do crescimento, retardo de crescimento, aparência facial típica (testa proeminente, olhos protuberantes, nariz fino com ponta em bico, lábios finos, micrognatia e orelhas salientes) e características dermatológicas distintas (alopecia generalizada, pele com aparência envelhecida, pele esclerótica e com covinhas no abdômen e extremidades, pele proeminente vasculatura, despigmentação, hipoplasia ungueal e perda de gordura subcutânea).
Escala de raridade
<1/50kMuito rara
1/20kRara
1/10kPouco freq.
1/5kIncomum
1/2k
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Partes do corpo afetadas
+ 35 sintomas em outras categorias
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 104 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Genes associados
2 genes identificados com associação a esta condição. Padrão de herança: Autosomal dominant, Autosomal recessive.
Transmembrane metalloprotease whose catalytic activity is critical for processing lamin A/LMNA on the inner nuclear membrane and clearing clogged translocons on the endoplasmic reticulum (PubMed:33293369, PubMed:33315887). Proteolytically removes the C-terminal three residues of farnesylated proteins (PubMed:33293369, PubMed:33315887). Also plays an antiviral role independently of its protease activity by restricting enveloped RNA and DNA viruses, including influenza A, Zika, Ebola, Sindbis, ves
Endoplasmic reticulum membraneNucleus inner membraneEarly endosome membraneLate endosome membrane
Mandibuloacral dysplasia with type B lipodystrophy
A form of mandibuloacral dysplasia, a rare progeroid disorder with clinical and genetic heterogeneity, characterized by growth retardation, craniofacial dysmorphic features due to distal bone resorption, musculoskeletal and skin abnormalities associated with lipodystrophy. MADB is a disease characterized by mandibular and clavicular hypoplasia, acroosteolysis, delayed closure of the cranial suture, joint contractures, and generalized lipodystrophy with loss of subcutaneous fat from the extremities, face, neck and trunk.
Lamins are intermediate filament proteins that assemble into a filamentous meshwork, and which constitute the major components of the nuclear lamina, a fibrous layer on the nucleoplasmic side of the inner nuclear membrane (PubMed:10080180, PubMed:10580070, PubMed:10587585, PubMed:10814726, PubMed:11799477, PubMed:12075506, PubMed:12927431, PubMed:15317753, PubMed:18551513, PubMed:18611980, PubMed:2188730, PubMed:22431096, PubMed:2344612, PubMed:23666920, PubMed:24741066, PubMed:31434876, PubMed:
Nucleus laminaNucleus envelopeNucleus, nucleoplasmNucleus matrixNucleus speckle
Emery-Dreifuss muscular dystrophy 2, autosomal dominant
A form of Emery-Dreifuss muscular dystrophy, a degenerative myopathy characterized by weakness and atrophy of muscle without involvement of the nervous system, early contractures of the elbows, Achilles tendons and spine, and cardiomyopathy associated with cardiac conduction defects.
Medicamentos aprovados (FDA)
1 medicamento encontrado nos registros da FDA americana.
Variantes genéticas (ClinVar)
967 variantes patogênicas registradas no ClinVar.
Classificação de variantes (ClinVar)
Distribuição de 7 variantes classificadas pelo ClinVar.
Vias biológicas (Reactome)
8 vias biológicas associadas aos genes desta condição.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Síndrome de progeria Hutchinson-Gilford
Selecione um estado ou use sua localização para ver resultados.
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Ensaios em destaque
🟢 Recrutando agora
1 pesquisa recrutando participantes. Converse com seu médico sobre a possibilidade de participar.
Outros ensaios clínicos
10 ensaios clínicos encontrados, 2 ativos.
Publicações mais relevantes
First Generation Proteolysis Targeting Chimeras (PROTACs) for the Treatment of Progeria.
Hutchinson-Gilford progeria syndrome (HGPS) is a rare genetic disorder caused by a mutation in the LMNA gene, leading to the production of progerin, an aberrant and toxic form of lamin A. Due to its hydrophobic nature, progerin accumulates at the nuclear membrane, disrupting nuclear architecture, impairing cellular functions, and ultimately resulting in death around adolescence. Reducing progerin levels is considered the most effective strategy to improve disease progression. To date, small-molecule approaches have primarily targeted progerin upstream mechanisms to indirectly reduce its levels. In this study, we report the development of first-generation proteolysis targeting chimeras (PROTACs) designed to directly degrade progerin, establishing a novel therapeutic paradigm for HGPS. We identify UCM-18142 (compound 2) as the first PROTAC capable of selectively degrading progerin. Treatment with UCM-18142 results in significant improvements in cellular phenotype in both HGPS patient-derived cells and a murine model, including enhanced proliferation, reduced senescence markers, and normalization of nuclear and mitochondrial abnormalities. Additionally, transcriptomic analysis of treated human cells reveals the cellular pathways modulated by compound. Remarkably, PROTAC 2 reduces progerin levels in vivo, supporting the therapeutic potential of this direct-targeting approach and opening new avenues for intervention in HGPS and related laminopathies.
Mitochondrial superoxide regulates nuclear envelope integrity and ageing via redox-mediated lipid metabolism.
The nuclear envelope (NE) is essential for cellular homeostasis, yet its integrity declines with age, accelerating functional deterioration. Here we report a mitochondria-to-NE signalling pathway that safeguards NE integrity through redox-dependent lipid metabolism. In Caenorhabditis elegans, reducing mitochondrial ETC activity preserves NE morphology during ageing. This effect requires developmental mitochondrial superoxide, which downregulates SBP-1 (SREBP orthologue) and suppresses unsaturated fatty acid biosynthesis. The resulting reduction in unsaturated fatty acid levels limits lipid peroxidation, thereby preserving NE structure. Interventions targeting lipid peroxidation preserve NE integrity, extend lifespan in worms and ameliorate senescence-associated phenotypes in human fibroblasts and monkey cells mimicking Hutchinson-Gilford progeria syndrome disease. Our findings reveal a previously unrecognized role for mitochondrial superoxide as a protective developmental signal that programs long-term NE integrity. This work establishes lipid peroxidation control as a conserved strategy to delay nuclear ageing and highlights redox-lipid cross-talk as a therapeutic axis for healthy ageing.
A study on the nutritional status and body composition of children with Hutchinson-Gilford progeria syndrome.
Hutchinson-Gilford Progeria Syndrome (HGPS) is a rare genetic disorder characterized by premature aging, severe growth retardation, and metabolic abnormalities. This study aimed to evaluate the growth, nutritional status, and body composition of HGPS patients, with a focus on fat and muscle distribution, to provide insights into potential nutritional and therapeutic interventions. Eight HGPS patients (aged ≥ 3 years) and 18 age- and sex-matched healthy controls were enrolled. Physical assessments, dietary surveys, and laboratory tests were conducted, including dual-energy X-ray absorptiometry (DXA) to analyze bone density, fat distribution, and muscle mass. Genetic testing confirmed LMNA mutations in all patients. Data on growth parameters, dietary intake, and metabolic profiles were collected and compared with controls. HGPS patients exhibited severe growth retardation, with significant declines in height and weight by two months of age compared to controls. Dietary surveys revealed that 4 out of 8 subjects had sufficient energy intake, the energy intake was 91 ± 39% of the estimated basal metabolic rate. DXA analysis showed reduced bone density (Z-score: -2.82 ± 1.46), abnormal fat distribution (increased visceral fat and decreased subcutaneous fat, the T/L fat ratio and A/G fat ratio > 95th percentile for boys and girls in China), and significant reductions in total and limb muscle mass(< mean-2SD). HGPS patients experience profound growth retardation, metabolic dysfunction, and abnormal body composition, including reduced muscle mass and altered fat distribution. A nutrient-dense diet with increased protein intake, healthy fat intake, and supplementation with vitamin D, calcium, and zinc are essential to support growth and muscle maintenance. Tailored and individualised dietary interventions may further improve outcomes. Continuous monitoring and further research are needed to optimize interventions and enhance the quality of life for HGPS patients.
RNA-coupled CRISPR screens reveal ZNF207 as a regulator of LMNA aberrant splicing in progeria.
Despite progress in understanding pre-mRNA splicing, the regulatory mechanisms controlling most alternative splicing events remain unclear. We developed CRASP-seq (CRISPR-based identification of regulators of alternative splicing with phenotypic sequencing), a method that integrates pooled CRISPR-based genetic perturbations with deep sequencing of splicing reporters, to quantitatively assess the impact of all human genes on alternative splicing from a single RNA sample. CRASP-seq identified both known and untested regulators, enriched for proteins involved in RNA splicing and metabolism. As a proof-of-concept, CRASP-seq analysis of the LMNA cryptic splicing event linked to progeria uncovered ZNF207, primarily known for mitotic spindle assembly, as a regulator of progerin splicing. ZNF207 depletion enhances canonical LMNA splicing and decreases progerin protein levels in patient-derived cells. We further show that ZNF207's zinc-finger domain broadly impacts alternative splicing through direct interactions with U1 small nuclear ribonucleoprotein (snRNP) components. These findings position ZNF207 as a U1 snRNP auxiliary factor and demonstrate the power of CRASP-seq to uncover key regulators and domains of alternative splicing.
Lipid Metabolism Alterations in Hereditary Inorganic Pyrophosphate Deficiency Syndromes: A Narrative Review of Insights and Controversies.
Pathological ectopic calcification of soft tissues can arise from reduced or absent levels of inorganic pyrophosphate (PPi), a key inhibitor of calcium hydroxyapatite deposition in soft connective tissues. The role of PPi in regulating mineralization has been recognized for decades, thanks to the pivotal work of Herbert Fleisch and colleagues; and its clinical relevance has been underscored by the identification of hereditary metabolic disorders, collectively termed PPi deficiency syndromes. These are caused by pathogenic variants in the essential genes for maintaining PPi homeostasis: ATP-binding cassette subfamily C member 6 (ABCC6), ectonucleotide pyrophosphate phosphodiesterase 1 (ENPP1), progressive ankylosis protein (ANK), tissue-nonspecific alkaline phosphatase (ALPL), CD73, and CD39. In recent years, abnormalities in lipid metabolism have been reported in these monogenic conditions. However, a common understanding of these alterations has yet to be established. This review provides an overview of the pathophysiology of PPi deficiency syndromes-pseudoxanthoma elasticum, generalized arterial calcification of infancy, arterial calcification due to CD73 deficiency, ankylosis, and Hutchinson-Gilford progeria syndrome-highlighting the lipid metabolism alterations in cells, animal models, and patients. We explore the evidence for a potential role of PPi-regulating proteins in lipid metabolic pathways to demonstrate that lipid alterations are not coincidental but entail opportunities for future research and for potential therapeutic interventions.
Publicações recentes
Mapping the Landscape of Hutchinson-Gilford Progeria Syndrome Research: A Bibliometric Analysis (1995-2025).
Modeling premature aging in yeast via the expression of Progerin.
Murine Progeria Model Exhibits Delayed Fracture Healing With Senescent Phenotype and Dysregulated Immune Response.
Epidural hematoma in a pediatric patient with Hutchinson-Gilford progeria syndrome: management considerations: a case report.
STING causes replication stress and nascent DNA degradation via SAMHD1.
📚 EuropePMC393 artigos no totalmostrando 194
First Generation Proteolysis Targeting Chimeras (PROTACs) for the Treatment of Progeria.
Advanced science (Weinheim, Baden-Wurttemberg, Germany)Hutchinson-Gilford progeria syndrome alters the endothelial genetic response to laminar shear stress.
Frontiers in physiologyProgerin expression in humans: Implications for natural ageing.
Mechanisms of ageing and developmentGeneral anesthesia in patient with Hutchinson-Gilford Progeria syndrome: two case reports of dental treatment in the one patient.
BMC anesthesiologyKRAS Can Bind to FTase Despite Disruption of the CAAX Binding Site.
BiochemistryMitochondrial superoxide regulates nuclear envelope integrity and ageing via redox-mediated lipid metabolism.
Nature metabolismStem cell-associated osteogenic deficiency causes craniofacial deformities with progeroid accumulation of prelamin A.
JCI insightA study on the nutritional status and body composition of children with Hutchinson-Gilford progeria syndrome.
Orphanet journal of rare diseasesCraniofacial features associated with Hutchinson - Gilford progeria syndrome - A case report.
StomatologijaSenescence-inhibitory Δ133p53α counteracts accelerated ageing and mortality.
bioRxiv : the preprint server for biologyTranscriptional profiling of Hutchinson-Gilford progeria patients identifies primary target pathways of progerin.
Nucleus (Austin, Tex.)RNA-coupled CRISPR screens reveal ZNF207 as a regulator of LMNA aberrant splicing in progeria.
Molecular cellWhy and How Are Infants with Hutchinson-Gilford Progeria Syndrome Born Without Severe Manifestations?
Medical sciences (Basel, Switzerland)Lipid Metabolism Alterations in Hereditary Inorganic Pyrophosphate Deficiency Syndromes: A Narrative Review of Insights and Controversies.
Journal of inherited metabolic diseaseDHT ameliorates cardiac aging in progeroid mice by XRCC4-mediated genome stabilization.
Mechanisms of ageing and developmentLonafarnib Clinical Trials Demonstrate Uncoupling of the Muscle-Bone Unit in Hutchinson-Gilford Progeria Syndrome.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchThe unfolded protein response in progeria arteries originates from non-endothelial cell types.
Life science allianceAuthor Correction: p300 nucleocytoplasmic shuttling underlies mTORC1 hyperactivation in Hutchinson-Gilford progeria syndrome.
Nature cell biologyVascular organoid model of Hutchinson-Gilford progeria syndrome uncovers repression of the SRF pathway in premature aging.
Developmental cellMicrohand Platform Equipped with Plate-Shaped End-Effectors Enables Precise Probing of Intracellular Structure Contribution to Whole-Cell Mechanical Properties.
MicromachinesSingle-cell RNA sequencing analysis reveals the critical role of fibroblasts in aortic progeria-associated vascular remodeling in Hutchinson-Gilford progeria syndrome mice.
Frontiers in immunologyAdvances of pharmacological therapies in lipodystrophy syndromes: current evidence and future directions.
Expert review of endocrinology & metabolismGeneration of Nonintegrative-Induced Pluripotent Stem Cells in Hutchinson-Gilford Progeria Syndrome: Enhancing Aging Research.
Aging medicine (Milton (N.S.W))miR-140-5p Overexpression Contributes to Oxidative Stress and Mitochondrial Dysfunction in Hutchinson-Gilford Progeria Syndrome Fibroblasts Through NRF2 Pathway.
Aging cellSelection of specific and efficient siRNAs in new cellular model for Hutchinson-Gilford progeria syndrome therapy.
Molecular therapy. Nucleic acidsBaricitinib Augments Lonafarnib Therapy to Preserve Colonic Homeostasis and Microbial Balance in a Mouse Model of Progeria.
Aging cellPOLR3A mutations cause nucleolus abnormalities and aberrant telomerase RNA metabolism in induced pluripotent stem cells from Wiedemann-Rautenstrauch premature aging syndrome patient.
BiogerontologyRare case of longevity in Hutchinson-Gilford progeria syndrome and literature review.
Orphanet journal of rare diseasesUnraveling the mysteries of Hutchinson-Gilford progeria syndrome: a comprehensive review of LMNA gene mutations.
BiogerontologyPharmacologic activation of Δ133p53α reduces cellular senescence in progeria patients-derived cells.
Aging pathobiology and therapeuticsPrelamin A Does Not Promote Atherosclerosis or Vascular Smooth Muscle Loss.
bioRxiv : the preprint server for biologyThe Essence of Nature Can be the Simplest (6)-Lifespan: Determined by Extracellular Fenton Chemistry.
Chemistry & biodiversityCounteracting lysosome defects alleviates the cellular senescence of Hutchinson-Gilford progeria syndrome.
Science China. Life sciencesSurgical Aortic Valve Replacement Combined With Coronary Artery Bypass Grafting in a Patient With Progeria.
JACC. Case reportsAnalysis of Beta-Dystroglycan in Different Cell Models of Senescence.
International journal of molecular sciencesDeregulated miR-145 and miR-27b in Hutchinson-Gilford progeria syndrome: implications for adipogenesis.
AgingImpact of miR-181a on SIRT1 Expression and Senescence in Hutchinson-Gilford Progeria Syndrome.
Diseases (Basel, Switzerland)Therapeutic RNA cleavage with RfxCas13d in Hutchinson-Gilford progeria syndrome.
Molecular therapy : the journal of the American Society of Gene TherapyThe senescence-inhibitory p53 isoform Δ133p53α: enhancing cancer immunotherapy and exploring novel therapeutic approaches for senescence-associated diseases.
GeroScienceA Quantitative High-Throughput Screen Identifies Compounds that Upregulate the p53 Isoform Δ133p53α and Inhibit Cellular Senescence.
ACS pharmacology & translational scienceNicotinamide Mononucleotide Alleviates Aging Defects in Hutchinson-Gilford Progeria Syndrome.
FASEB journal : official publication of the Federation of American Societies for Experimental BiologyThe accumulation of progerin underlies the loss of aortic smooth muscle cells in Hutchinson-Gilford progeria syndrome.
Cell death & diseaseInvolvement of NRF2 and AMPK signaling in aging and progeria: a digest.
Redox biologyNational survey of Hutchinson-Gilford progeria syndrome and progeroid laminopathy in Japan.
AgingA noncanonical cGAS-STING pathway drives cellular and organismal aging.
Proceedings of the National Academy of Sciences of the United States of AmericaPatient-Derived Cortical Organoids Reveal Senescence of Neural Progenitor Cells in Hutchinson-Gilford Progeria Syndrome.
Aging cellAssessing the Efficacy of Small Molecule Drugs in Hutchinson-Gilford Progeria Syndrome: A Review of Clinical Trials.
Reviews on recent clinical trialsPrecise progerin targeting using RfxCas13d: A therapeutic avenue for Hutchinson-Gilford progeria syndrome.
Molecular therapy : the journal of the American Society of Gene TherapyRecurrent somatic mutation and progerin expression in early vascular aging of chronic kidney disease.
Nature agingPrecision Reprogramming-Restoring Function to Aged Cells.
Cellular reprogrammingRole of Lysophosphatidic Acid in Neurological Diseases: From Pathophysiology to Therapeutic Implications.
Frontiers in bioscience (Landmark edition)Investigating telomere length in progeroid syndromes: implications for aging disorders.
AgingBaricitinib and Lonafarnib Synergistically Target Progerin and Inflammation, Improving Lifespan and Health in Progeria Mice.
International journal of molecular sciencesStrategies for improved endothelial cell adhesion in microphysiological vascular model systems.
PloS oneHutchinson-Gilford Progeria Syndrome.
JAMA dermatologyAttenuated Nuclear Tension Regulates Progerin-Induced Mechanosensitive Nuclear Wrinkling and Chromatin Remodeling.
Advanced science (Weinheim, Baden-Wurttemberg, Germany)Phase behavior and dissociation kinetics of lamins in a polymer model of progeria.
The Journal of chemical physicsMesenchymal Stem Cell Therapy for Hutchinson-Gilford Progeria: Improvements in Arterial Stiffness and Bone Mineral Density in a Single Case.
Children (Basel, Switzerland)Sclerotic skin in Hutchinson-Gilford progeria syndrome: Reversible senescent phenotype.
Journal der Deutschen Dermatologischen Gesellschaft = Journal of the German Society of Dermatology : JDDGAnesthetic management of a child with Hutchinson-Gilford progeria syndrome.
Journal of anaesthesiology, clinical pharmacologyVascular cell types in progeria: victims or villains?
Trends in molecular medicineA non-canonical cGAS-STING pathway drives cellular and organismal aging.
bioRxiv : the preprint server for biologyAuthor Correction: Recapitulation of premature ageing with iPSCs from Hutchinson-Gilford progeria syndrome.
NatureDisease pathogenicity in Hutchinson-Gilford progeria syndrome mice: insights from lung-associated alterations.
Molecular medicine (Cambridge, Mass.)Adenine base editing rescues pathogenic phenotypes in tissue engineered vascular model of Hutchinson-Gilford progeria syndrome.
APL bioengineeringIntracranial hypertension in a patient with Hutchinson-Gilford progeria syndrome.
Archives de pediatrie : organe officiel de la Societe francaise de pediatrieIndirect bypass for revascularization in Hutchinson-Gilford progeria syndrome: an illustrative case.
Child's nervous system : ChNS : official journal of the International Society for Pediatric NeurosurgeryMetformin delays the decline in thermogenic function of brown adipose tissue in a mouse model of Hutchinson-Gilford progeria syndrome.
Experimental gerontologyTargeting CRM1 for Progeria Syndrome Therapy.
Aging cellThe anti-senescence effect of D-β-hydroxybutyrate in Hutchinson-Gilford progeria syndrome involves progerin clearance by the activation of the AMPK-mTOR-autophagy pathway.
GeroScienceLongitudinal Changes in Myocardial Deformation in Hutchinson-Gilford Progeria Syndrome.
Circulation. Cardiovascular imagingThe role of the LINC complex in ageing and microgravity.
Mechanisms of ageing and developmentAngiopoietin-2: A Therapeutic Target for Vascular Protection in Hutchinson-Gilford Progeria Syndrome.
International journal of molecular sciencesVascular dysfunction in Hutchinson-Gilford progeria syndrome.
Trends in molecular medicineAdvances of NAT10 in diseases: insights from dual properties as protein and RNA acetyltransferase.
Cell biology and toxicologyHutchinson-Gilford progeria syndrome: unraveling the genetic basis, symptoms, and advancements in therapeutic approaches.
Therapeutic advances in rare diseaseRoles of the lamin A-specific tail region in the localization to sites of nuclear envelope rupture.
PNAS nexusProgerin can induce DNA damage in the absence of global changes in replication or cell proliferation.
PloS oneAn Analysis of Hearing Outcomes in Children with Hutchinson-Gilford Progeria Syndrome.
British journal of hospital medicine (London, England : 2005)Characterization of the craniofacial abnormalities of the homozygous G608G progeria mouse model.
Frontiers in physiologyReactivation of senescence-associated endogenous retroviruses by ATF3 drives interferon signaling in aging.
Nature agingEndothelial cell-specific progerin expression does not cause cardiovascular alterations and premature death.
Aging cellEnhancing Cellular Homeostasis: Targeted Botanical Compounds Boost Cellular Health Functions in Normal and Premature Aging Fibroblasts.
BiomoleculesGeneration of a lamin A/C knockout human induced pluripotent stem cell line (ZJULLi007-A) via CRISPR/Cas9.
Stem cell researchAngiopoietin-2 reverses endothelial cell dysfunction in progeria vasculature.
Aging cellRedefining Aging: A Tale of Hutchinson-Gilford Progeria Syndrome.
The Journal of the Association of Physicians of IndiaExpansion in situ genome sequencing links nuclear abnormalities to hotspots of aberrant euchromatin repression.
bioRxiv : the preprint server for biologyNuclear envelope budding inhibition slows down progerin-induced aging process.
Proceedings of the National Academy of Sciences of the United States of AmericaEndothelial YAP/TAZ activation promotes atherosclerosis in a mouse model of Hutchinson-Gilford progeria syndrome.
The Journal of clinical investigationInhibition of poly(ADP-Ribosyl)ation reduced vascular smooth muscle cells loss and improves aortic disease in a mouse model of human accelerated aging syndrome.
Cell death & diseaseInflammation and Fibrosis in Progeria: Organ-Specific Responses in an HGPS Mouse Model.
International journal of molecular sciencesLipodystrophic Laminopathies: From Dunnigan Disease to Progeroid Syndromes.
International journal of molecular sciencesTargeted partial reprogramming of age-associated cell states improves markers of health in mouse models of aging.
Science translational medicineMechanotransduction of the vasculature in Hutchinson-Gilford Progeria Syndrome.
Frontiers in physiologyProgerin mRNA expression in non-HGPS patients is correlated with widespread shifts in transcript isoforms.
NAR genomics and bioinformaticsA novel role for CSA in the regulation of nuclear envelope integrity: uncovering a non-canonical function.
Life science allianceEndothelial-to-Mesenchymal Transition Contributes to Accelerated Atherosclerosis in Hutchinson-Gilford Progeria Syndrome.
CirculationEnd-stage kidney disease treated with hemodialysis in a patient with Hutchinson-Gilford progeria syndrome.
Polish archives of internal medicinePremature aging effects on COVID-19 pathogenesis: new insights from mouse models.
Scientific reportsAnesthetic Management of Cardiopulmonary Bypass in Hutchinson-Gilford Progeria Syndrome: A Case Report.
A&A practiceNavigating Lipodystrophy: Insights from Laminopathies and Beyond.
International journal of molecular sciencesA Novel Role for the Longevity-Associated Protein SLC39A11 as a Manganese Transporter.
Research (Washington, D.C.)Aberrant migration features in primary skin fibroblasts of Huntington's disease patients hold potential for unraveling disease progression using an image based machine learning tool.
Computers in biology and medicineA Review of Research on the Mechanism of Tumor Regulation by N-Acetyltransferase 10.
Discovery medicinemtDNA release promotes cGAS-STING activation and accelerated aging of postmitotic muscle cells.
Cell death & diseaseHutchinson-Gilford progeria syndrome mice display accelerated arterial thrombus formation and increased platelet reactivity.
Thrombosis researchCardiac and skeletal muscle manifestations in the G608G mouse model of Hutchinson-Gilford progeria syndrome.
Aging cellA Pair of Sibling Patients With Premature Aging Syndrome of Unknown Etiology.
CureusProgerin forms an abnormal meshwork and has a dominant-negative effect on the nuclear lamina.
Proceedings of the National Academy of Sciences of the United States of AmericaMurine Progeria Model Exhibits Delayed Fracture Healing with Dysregulated Local Immune Response.
bioRxiv : the preprint server for biologyReticulate sclerodermoid pigmentation in Hutchinson-Gilford progeria syndrome - A report of two cases.
Indian journal of dermatology, venereology and leprologyThe Use of Peripheral Nerve Block and Intra-articular Steroid Injection for Pain Management in an Adolescent with Hutchinson-Gilford Progeria Syndrome: A Case Report.
Acta medica PhilippinaLife at the crossroads: the nuclear LINC complex and vascular mechanotransduction.
Frontiers in physiologyPathogenic hyperactivation of mTORC1 by cytoplasmic EP300 in Hutchinson-Gilford progeria syndrome.
Cell stressIntervention for critical aortic stenosis in Hutchinson-Gilford progeria syndrome.
Frontiers in cardiovascular medicineDoxycycline decelerates aging in progeria mice.
Aging cellExacerbated atherosclerosis in progeria is prevented by progerin elimination in vascular smooth muscle cells but not endothelial cells.
Proceedings of the National Academy of Sciences of the United States of AmericaMolecular Basis for RNA Cytidine Acetylation by NAT10.
bioRxiv : the preprint server for biologyAged-vascular niche hinders osteogenesis of mesenchymal stem cells through paracrine repression of Wnt-axis.
Aging cellProgeria-based vascular model identifies networks associated with cardiovascular aging and disease.
Aging cellProgerin Inhibits the Proliferation and Migration of Melanoma Cells by Regulating the Expression of Paxillin.
OncoTargets and therapyGenetic and pharmacological modulation of lamin A farnesylation determines its function and turnover.
Aging cellMitophagy defect mediates the aging-associated hallmarks in Hutchinson-Gilford progeria syndrome.
Aging cellPremature aging in genetic diseases: what conclusions can be drawn for physiological aging.
Frontiers in agingRestoring functional TDP-43 oligomers in ALS and laminopathic cellular models through baicalein-induced reconfiguration of TDP-43 aggregates.
Scientific reportsAbnormal Myocardial Deformation Despite Normal Ejection Fraction in Hutchinson-Gilford Progeria Syndrome.
Journal of the American Heart Associationp300 nucleocytoplasmic shuttling underlies mTORC1 hyperactivation in Hutchinson-Gilford progeria syndrome.
Nature cell biologyEpidemiological characteristics of patients with Hutchinson-Gilford progeria syndrome and progeroid laminopathies in China.
Pediatric researchCRL2APPBP2-mediated TSPYL2 degradation counteracts human mesenchymal stem cell senescence.
Science China. Life sciencesRemodeling of the Cardiac Extracellular Matrix Proteome During Chronological and Pathological Aging.
Molecular & cellular proteomics : MCPThe farnesyl transferase inhibitor (FTI) lonafarnib improves nuclear morphology in ZMPSTE24-deficient fibroblasts from patients with the progeroid disorder MAD-B.
Nucleus (Austin, Tex.)Use of Farnesyl Transferase Inhibitors in an Ageing Model in Drosophila.
Journal of developmental biologyA new fluorescent probe for the visualization of progerin.
Bioorganic chemistryImpaired end joining induces cardiac atrophy in a Hutchinson-Gilford progeria mouse model.
Proceedings of the National Academy of Sciences of the United States of AmericaLong lifetime and tissue-specific accumulation of lamin A/C in Hutchinson-Gilford progeria syndrome.
The Journal of cell biologyAn infant with congenital micrognathia and upper airway obstruction was diagnosed as Hutchinson-Gilford progeria syndrome caused by a novel LMNA mutation: Case report and literature review.
HeliyonThe Activation of JAK/STAT3 Signaling and the Complement System Modulate Inflammation in the Primary Human Dermal Fibroblasts of PXE Patients.
BiomedicinesPrelamin A and ZMPSTE24 in premature and physiological aging.
Nucleus (Austin, Tex.)Development of an accelerated cellular model for early changes in Alzheimer's disease.
Scientific reportsGhrelin delays premature aging in Hutchinson-Gilford progeria syndrome.
Aging cellHutchinson-Gilford progeria syndrome: Cardiovascular manifestations and treatment.
Mechanisms of ageing and developmentProgerin, an Aberrant Spliced Form of Lamin A, Is a Potential Therapeutic Target for HGPS.
Cells[Progeroid syndromes : Aging, skin aging, and mechanisms of progeroid syndromes].
Dermatologie (Heidelberg, Germany)Hutchinson-Gilford progeria patient-derived cardiomyocyte model of carrying LMNA gene variant c.1824 C > T.
Cell and tissue researchAging Model for Analyzing Drug-Induced Proarrhythmia Risks Using Cardiomyocytes Differentiated from Progeria-Patient-Derived Induced Pluripotent Stem Cells.
International journal of molecular sciencesThe secretome atlas of two mouse models of progeria.
Aging cellImpact of Combined Baricitinib and FTI Treatment on Adipogenesis in Hutchinson-Gilford Progeria Syndrome and Other Lipodystrophic Laminopathies.
CellsProgeria Presenting with Pyogenic Granuloma in Conjunctiva: A Case Report.
JNMA; journal of the Nepal Medical AssociationHutchinson-Gilford Progeria Syndrome: Cellular Mechanisms and Therapeutic Perspectives.
Archives of medical researchA recurrent homozygous LMNA missense variant p.Thr528Met causes atypical progeroid syndrome characterized by mandibuloacral dysostosis, severe muscular dystrophy, and skeletal deformities.
American journal of medical genetics. Part ABone dysplasia in Hutchinson-Gilford progeria syndrome is associated with dysregulated differentiation and function of bone cell populations.
Aging cellHepatic hydrogen sulfide levels are reduced in mouse model of Hutchinson-Gilford progeria syndrome.
AgingA Safer Path to Cellular Rejuvenation: Endogenous Oct4 Activation via CRISPR/dCas9 in Progeria Mouse Models.
Cellular reprogrammingOnychodystrophy with Multiple Epiphyseal Dysplasia: Literature Review.
Skin appendage disordersAnaesthetic management of paediatric patient with Hutchinson-Gilford progeria syndrome: A case report.
Indian journal of anaesthesiaNeurovascular disease: 2022 update.
Free neuropathologyProgression of Cardiac Abnormalities in Hutchinson-Gilford Progeria Syndrome: A Prospective Longitudinal Study.
CirculationReadily Available Tools to Detect Progerin and Cardiac Disease Progression in Hutchinson-Gilford Progeria Syndrome.
CirculationCoronary and carotid artery dysfunction and KV7 overexpression in a mouse model of Hutchinson-Gilford progeria syndrome.
GeroScienceSenotherapeutic peptide treatment reduces biological age and senescence burden in human skin models.
npj agingAccelerated aging in articular cartilage by ZMPSTE24 deficiency leads to osteoarthritis with impaired metabolic signaling and epigenetic regulation.
Cell death & diseaseActivation of endoplasmic reticulum stress in premature aging via the inner nuclear membrane protein SUN2.
Cell reportsNuclear softening mediated by Sun2 suppression delays mechanical stress-induced cellular senescence.
Cell death discoveryProgerinin, an Inhibitor of Progerin, Alleviates Cardiac Abnormalities in a Model Mouse of Hutchinson-Gilford Progeria Syndrome.
CellsLong lifetime and selective accumulation of the A-type lamins accounts for the tissue specificity of Hutchinson-Gilford progeria syndrome.
bioRxiv : the preprint server for biologyBiomechanical and transcriptional evidence that smooth muscle cell death drives an osteochondrogenic phenotype and severe proximal vascular disease in progeria.
Biomechanics and modeling in mechanobiologyAuthor Correction: Unique progerin C-terminal peptide ameliorates Hutchinson-Gilford progeria syndrome phenotype by rescuing BUBR1.
Nature agingUnique progerin C-terminal peptide ameliorates Hutchinson-Gilford progeria syndrome phenotype by rescuing BUBR1.
Nature agingCorruption of DNA end-joining in mammalian chromosomes by progerin expression.
DNA repairThe Consideration of Pseudoxanthoma Elasticum as a Progeria Syndrome.
Frontiers in bioscience (Landmark edition)The Molecular and Cellular Basis of Hutchinson-Gilford Progeria Syndrome and Potential Treatments.
GenesAcute Coronary Syndrome Treated with Percutaneous Coronary Intervention in Hutchinson-Gilford Progeria.
Children (Basel, Switzerland)Lonafarnib and everolimus reduce pathology in iPSC-derived tissue engineered blood vessel model of Hutchinson-Gilford Progeria Syndrome.
Scientific reportsTranscriptional activation of endogenous Oct4 via the CRISPR/dCas9 activator ameliorates Hutchinson-Gilford progeria syndrome in mice.
Aging cellLonafarnib improves cardiovascular function and survival in a mouse model of Hutchinson-Gilford progeria syndrome.
eLifePlasma Progerin in Patients With Hutchinson-Gilford Progeria Syndrome: Immunoassay Development and Clinical Evaluation.
CirculationThe Senolytic Drug Fisetin Attenuates Bone Degeneration in the Zmpste24 -/- Progeria Mouse Model.
Journal of osteoporosisPerturbed actin cap as a new personalized biomarker in primary fibroblasts of Huntington's disease patients.
Frontiers in cell and developmental biologySmooth Muscle Cell Death Drives an Osteochondrogenic Phenotype and Severe Proximal Vascular Disease in Progeria.
bioRxiv : the preprint server for biologyRescue of Mitochondrial Function in Hutchinson-Gilford Progeria Syndrome by the Pharmacological Modulation of Exportin CRM1.
CellsNuclear envelope morphology change upon repetitive treatment with modified antisense oligonucleotides targeting Hutchinson-Gilford Progeria Syndrome.
Biochemistry and biophysics reportsBaseline Range of Motion, Strength, Motor Function, and Participation in Youth with Hutchinson-Gilford Progeria Syndrome.
Physical & occupational therapy in pediatricsSTAT1 Drives the Interferon-Like Response and Aging Hallmarks in Progeria.
Aging biologyTranscriptional profiling of Hutchinson-Gilford Progeria syndrome fibroblasts reveals deficits in mesenchymal stem cell commitment to differentiation related to early events in endochondral ossification.
eLifeDetection of Cerebrovascular Disease in a Child with Hutchinson-Gilford Progeria Syndrome Using MR Angiography: A Case Report.
Journal of the Korean Society of RadiologyHutchinson-Gilford progeria syndrome complicated with stroke: A report of 2 cases and literature review.
Frontiers in pediatricsFDA approval summary for lonafarnib (Zokinvy) for the treatment of Hutchinson-Gilford progeria syndrome and processing-deficient progeroid laminopathies.
Genetics in medicine : official journal of the American College of Medical GeneticsModelling premature cardiac aging with induced pluripotent stem cells from a hutchinson-gilford Progeria Syndrome patient.
Frontiers in physiologyMitochondrial Dysfunction and Oxidative Stress in Hereditary Ectopic Calcification Diseases.
International journal of molecular sciencesProgerin induces a phenotypic switch in vascular smooth muscle cells and triggers replication stress and an aging-associated secretory signature.
GeroScienceThe role of prelamin A post-translational maturation in stress response and 53BP1 recruitment.
Frontiers in cell and developmental biologyInhibition of glutaminolysis restores mitochondrial function in senescent stem cells.
Cell reportsStructural basis for the interaction between unfarnesylated progerin and the Ig-like domain of lamin A/C in premature aging disorders.
Biochemical and biophysical research communicationsThe Heterochromatin protein 1 is a regulator in RNA splicing precision deficient in ulcerative colitis.
Nature communicationsAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Síndrome de progeria Hutchinson-Gilford.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Síndrome de progeria Hutchinson-Gilford
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- First Generation Proteolysis Targeting Chimeras (PROTACs) for the Treatment of Progeria.
- Mitochondrial superoxide regulates nuclear envelope integrity and ageing via redox-mediated lipid metabolism.
- A study on the nutritional status and body composition of children with Hutchinson-Gilford progeria syndrome.
- RNA-coupled CRISPR screens reveal ZNF207 as a regulator of LMNA aberrant splicing in progeria.
- Lipid Metabolism Alterations in Hereditary Inorganic Pyrophosphate Deficiency Syndromes: A Narrative Review of Insights and Controversies.
- Mapping the Landscape of Hutchinson-Gilford Progeria Syndrome Research: A Bibliometric Analysis (1995-2025).
- Modeling premature aging in yeast via the expression of Progerin.
- Murine Progeria Model Exhibits Delayed Fracture Healing With Senescent Phenotype and Dysregulated Immune Response.
- Epidural hematoma in a pediatric patient with Hutchinson-Gilford progeria syndrome: management considerations: a case report.
- STING causes replication stress and nascent DNA degradation via SAMHD1.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:740(Orphanet)
- OMIM OMIM:176670(OMIM)
- MONDO:0008310(MONDO)
- GARD:7467(GARD (NIH))
- Variantes catalogadas(ClinVar)
- Busca completa no PubMed(PubMed)
- Artigo Wikipedia(Wikipedia)
- Q213098(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar