Introdução
O que você precisa saber de cara
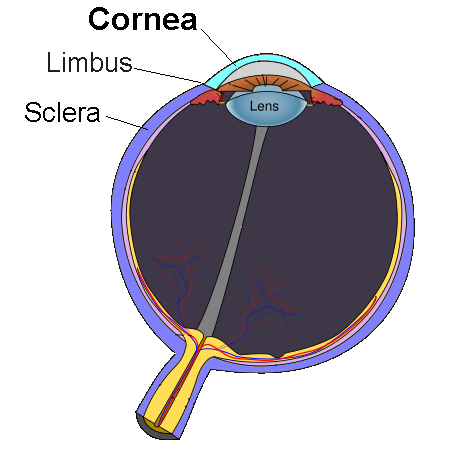
A distrofia da membrana basal epitelial (EBMD) é um distúrbio ocular que pode causar dor e ressecamento. A EBMD, também conhecida como distrofia mapa-ponto-impressão digital e distrofia epitelial microcística de Cogan, é uma doença epitelial da córnea que pode resultar em erosões corneanas recorrentes, astigmatismo corneano irregular e diminuição da visão.
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 8 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Genes associados
1 gene identificado com associação a esta condição.
Plays a role in cell adhesion (PubMed:8024701). May play a role in cell-collagen interactions (By similarity)
SecretedSecreted, extracellular space, extracellular matrix
Corneal dystrophy, epithelial basement membrane
A bilateral anterior corneal dystrophy characterized by grayish epithelial fingerprint lines, geographic map-like lines, and dots (or microcysts) on slit-lamp examination. Pathologic studies show abnormal, redundant basement membrane and intraepithelial lacunae filled with cellular debris.
Variantes genéticas (ClinVar)
39 variantes patogênicas registradas no ClinVar.
Classificação de variantes (ClinVar)
Distribuição de 8 variantes classificadas pelo ClinVar.
Vias biológicas (Reactome)
1 via biológica associada aos genes desta condição.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Distrofia corneana, microcística
Selecione um estado ou use sua localização para ver resultados.
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Ensaios em destaque
Pesquisa e ensaios clínicos
5 ensaios clínicos encontrados, 1 ativos.
Publicações mais relevantes
Meesmann Corneal Dystrophy with Epithelial Basement Membrane Abnormalities: Clinical and Genetic Analysis of Two Families with Novel and Known Mutations in KRT3 and KRT12.
This study describes the clinical and genetic features of Meesmann epithelial corneal dystrophy (MECD) in two unrelated families and reports new genotype-phenotype associations. Ten patients from a Lebanese family (n = 4) (Family 1) and a Spanish family (n = 6) (Family 2) underwent ophthalmologic evaluation, in vivo confocal microscopy (IVCM), anterior segment optical coherence tomography (AS-OCT) with epithelial thickness mapping (ET-map), and targeted next-generation sequencing (NGS) using a custom-designed 133-gene panel associated with anterior segment dystrophies. In Family 1, a novel homozygous KRT12 c.1181T>C (p.Leu394Pro) variant was identified in the symptomatic proband and his clinically asymptomatic brother, while both parents, who were first cousins, were heterozygous for this nucleotide variant. The proband also carried the heterozygous KRT3 c.250C>T (p.Arg84Trp) variant, which has been previously reported but, to our knowledge, has not been described in co-occurrence until now. In addition, the proband showed a complex phenotype with signs of MECD and epithelial basal membrane alterations consistent with epithelial basement membrane dystrophy (EBMD). In Family 2, four affected members carried the KRT3 c.1492G>A (p.Glu498Lys) variant in heterozygosity, which has been previously described. The elderly members affected showed typical signs of MECD and EBMD. To our knowledge, these concomitant alterations have not been previously described with genetical confirmation. In conclusion, this study provides the first evidence that the co-occurrence of variants in two Meesmann corneal dystrophy-associated genes (KRT3 and KRT12) can jointly account for the disease phenotype. We also highlight the association of MECD with EBMD in both families. Characterization using IVCM and AS-OCT ET-Map provides a deeper understanding of the morphological changes and phenotypic variability in MECD, confirming the utility of this multimodal imaging approach for diagnosis and management.
Superficial Keratectomy with Cryopreserved Amniotic Membrane for Ocular Surface Optimization in Patients with Epithelial Basement Membrane Dystrophy.
Epithelial Basement Membrane Dystrophy (EBMD)-induced irregular astigmatism can lead to unreliable biometry and suboptimal refractive outcomes in cataract surgery patients. Herein, we assessed the use of superficial keratectomy followed by placement of cryopreserved amniotic membrane (cAM) to improve the ocular surface. This was a single-center, retrospective study of consecutive patients with EBMD that underwent superficial keratectomy with adjunctive treatment with cAM between November 2022 and March 2025. Time to complete epithelialization, visual acuity (LogMAR), higher order aberration Root Mean Square (HOA RMS), cylinder power, and IOL biometry were assessed up to two months post-treatment. A total of 27 eyes of 21 patients with EBMD were included in the study and received cAM for a mean of 3.0 ± 0.4 days. Complete re-epithelialization was noted in an average 7.0 ± 4.7 days, with 96.3% (26/27) of eyes achieved epithelialization within 10 days. At the two month follow up, LogMAR VA slightly improved from 0.34 ± 0.21 to 0.31 ± 0.23 (p=0.42). HOA RMS significantly improved from 0.68 ± 0.53 μm to 0.42 ± 0.23 μm (p=0.006), and cylinder power significantly changed from 1.38 ± 0.75 D to 1.09 ± 0.71 D (p=0.022). Superficial keratectomy with cAM led to complete re-epithelialization in the majority (96.3%) of cases within 10 days, which was associated with a significant change in HOA RMS, cylinder power and IOL SE. This treatment may be considered in patients prior to cataract surgery to return the cornea to healthy state and change the biometry input for IOL selection.
Analysis of corneal wavefront aberrations and corneal densitometry in eyes with epithelial basement membrane dystrophy.
To investigate wavefront aberrations, as well as corneal optical densitometry (COD), in eyes with epithelial basement membrane dystrophy (EBMD) and the influence on visual acuity. In this cross-sectional study, 70 eyes of 70 patients (mean age 55.9 ± 14.0 years) with the central cornea involving EBMD were compared to 50 healthy eyes of 50 patients (mean age 58.8 ± 14.1 years) serving as controls. Wavefront aberrations of the anterior corneal surface and the total cornea were measured with the Pentacam AXL (Oculus Optikgeräte GmbH, Wetzlar, Germany), and calculated for the 6 mm central corneal zone. In addition, the COD (corneal light backscatter measured in grey scale units) of the anterior 120 µm of the central 0-2 mm, 2-6 mm, and 6-10 mm of the cornea was evaluated. Corrected distance visual acuity (CDVA) was correlated with wavefront aberrations and COD using Spearman correlation analysis. EBMD resulted in significant higher peak-to-valley (PTV; median: 15.0 [interquartile range: 9] µm), square root of the sum of the squared higher-order aberrations (RMS-HOA; 0.77 [0.52] µm), astigmatism (1.06 [1.04] µm), coma (0.41 [0.44] µm), and trefoil (0.28 [0.40] µm) (all p ≤ 0.01). A moderate correlation was found especially between CDVA and PTV as well as RMS-HOA. EBMD led to a statistically significant higher COD (p < 0.01) in the central corneal 6-mm and correlated moderately with CDVA outcomes. Our study revealed a significant correlation between elevated wavefront aberrations and backscattering in eyes affected by epithelial basement membrane dystrophy. While COD demonstrates potential for diagnostic purposes, additional studies are necessary to ascertain its specificity and distinguish EBMD from other ocular surface disorders.
Concurrent Meesmann Corneal Dystrophy and Epithelial Basement Membrane Dystrophy.
Salzmann nodular corneal degeneration (SNCD) is a relatively rare, slowly progressive, non-inflammatory condition of the cornea that is often underrecognized and frequently misdiagnosed due to its subtle early manifestations and overlapping features with other corneal disorders. In 1925, Austrian ophthalmologist Maximilian Salzmann described a series of patients with bluish-white corneal nodules secondary to phlyctenular or trachomatous keratitis. Although initially termed as Salzmann nodular corneal dystrophy, SNCD was later redefined as an indolent, degenerative condition. SNCD is characterised by the formation of fibrocellular outgrowth anterior to the basement membrane, resulting in elevated nodules most commonly involving the mid-periphery of the cornea. The disease was initially thought to be a variant of corneal dystrophy associated with phlyctenular and trachomatous keratitis. However, as clinical and histopathological understanding evolved, the term Salzmann nodular dystrophy was refined to Salzmann nodular degeneration to better reflect its acquired, degenerative nature. SNCD may be either primary or secondary. Primary Salzmann, the most common form, is typically not associated with any other ocular surface disease. However, these patients typically have coexisting meibomian gland dysfunction. Infrequently, SNCD has been observed in cases with chronic ocular surface inflammation, such as vernal keratoconjunctivitis, trachoma, interstitial keratitis, and dry eyes, as well as in non-inflammatory conditions, including epithelial basement membrane dystrophy and chronic rigid contact lens use. SNCD typically occurs in the fifth to sixth decade of life, though it has been reported in individuals aged 4 to 90. Although most cases are bilateral, it may present asymmetrically. SNCD predominantly affects women and may affect the White population more often. Spontaneous remission is unlikely, but symptoms typically improve with lubrication and topical anti-inflammatory therapy. Surgical options in refractory cases include manual excision, phototherapeutic keratectomy (PTK) with or without the use of topical mitomycin-C, and lamellar or penetrating keratoplasty. Though many patients are asymptomatic, SNCD may cause significant morbidity and contribute to the degradation of vision. The prognosis is generally favorable; however, prompt recognition and management of SNCD, along with its underlying etiologies, is critical, as no cases of spontaneous remission have been reported. SNCD is characterized by the development of discrete, elevated, bluish-white, superficial nodules, typically situated in the mid-peripheral or peripheral cornea. These nodules consist of collagenous fibrocellular material deposited anterior to Bowman's layer and the epithelial basement membrane. The overlying epithelium remains intact but may show irregularity or thinning. The condition is often bilateral but asymmetrical and may be either primary (idiopathic) or secondary to chronic ocular surface inflammation or trauma. To understand the pathogenesis of SNCD, it is essential to appreciate the anatomical structure of the cornea. The cornea is composed of 5 primary layers—the epithelium, Bowman's layer, stroma, Descemet's membrane, and endothelium. The epithelial basement membrane, situated beneath the epithelium, plays a crucial role in maintaining corneal homeostasis and facilitating repair. In SNCD, pathology predominantly occurs in the anterior cornea, with fibrocellular deposits accumulating between the epithelial basement membrane and Bowman's layer, or replacing Bowman's layer altogether. Histologically, the nodules in SNCD demonstrate replacement of Bowman's layer by extracellular matrix material, including collagen types I and III, and are often accompanied by activated keratocytes and myofibroblasts. This process reflects a reparative or reactive response to chronic epithelial stress, inflammation, or trauma, rather than a genetically determined dystrophy. This distinction is important, as SNCD is not inherited and does not follow a predictable pattern of progression typical of true dystrophies. SNCD can be broadly categorized into 2 types—primary (idiopathic) and secondary. In primary SNCD, there is no identifiable preceding ocular pathology. Primary SNCD typically presents in middle-aged women and may be associated with coexisting meibomian gland dysfunction or ocular surface dryness, although not directly caused by any specific ocular condition. This form is more common and typically arises without an apparent trigger. In contrast, secondary SNCD develops in the context of chronic ocular surface disease or corneal structural abnormalities. Several ocular conditions have been implicated in its pathogenesis, including vernal keratoconjunctivitis, trachoma, interstitial keratitis, and chronic blepharitis. Patients with severe dry eye disease or limbal stem cell deficiency are also at elevated risk. Other predisposing factors include recurrent epithelial erosions, a history of ocular trauma or surgery, and long-term contact lens wear, all of which contribute to chronic irritation and corneal remodeling. Additionally, epithelial basement membrane dystrophy has been associated with SNCD, likely due to persistent epithelial irregularity and defective wound healing responses. Understanding the classification and associated conditions of SNCD is essential for targeted management and prevention of recurrence. The pathophysiology likely involves chronic low-grade inflammation and recurrent epithelial microtrauma, leading to aberrant healing and subepithelial fibrosis. The exact molecular mechanisms are not yet fully understood, but they involve dysregulated wound healing, the activation of matrix metalloproteinases (MMPs), and the stimulation of fibroblasts. SNCD typically manifests in individuals aged 40 to 70, though cases have been reported in children as young as 4 and adults older than 90. SNCD shows a clear female predominance and is more frequently reported among the Caucasian population. The condition is bilateral in over 60% of cases, but it may present with highly asymmetrical features. Due to its often indolent course, many cases remain undiagnosed unless the nodules affect the visual axis or cause ocular discomfort. Patients with SNCD may remain asymptomatic, particularly in the early stages or when the nodules are located outside the visual axis, thus sparing the central vision. However, as the disease progresses or when the nodules encroach upon the optical zone, a variety of symptoms may develop. A common complaint is a persistent foreign body sensation, often caused by elevated nodules that disrupt the smooth corneal surface. Decreased visual acuity can result from direct obstruction of the visual axis or secondary to irregular corneal astigmatism induced by the nodules. Patients may also experience fluctuating vision, particularly in association with tear film instability. Photophobia and ocular irritation are frequently reported, especially when there is concomitant ocular surface inflammation. In some cases, blurred vision may be exacerbated by surface irregularities that alter the cornea's refractive contour. These symptoms collectively impact the patient's visual function and quality of life, emphasizing the importance of timely diagnosis and tailored management. The nodules are typically 1 to 3 mm in diameter, round to oval in shape, firm, and elevated. These nodules often appear bluish-white due to underlying stromal haze and are most frequently located in the midperipheral or superior cornea. In some cases, central involvement can significantly impair visual function. Diagnosis is primarily clinical, based on slit-lamp biomicroscopy. Nodules appear elevated, smooth, and opalescent. Adjacent corneal thinning or superficial vascularization may occasionally be observed. Corneal topography may show irregular astigmatism, and anterior segment optical coherence tomography (AS-OCT) can confirm subepithelial location, epithelial thinning, and loss of Bowman's layer. IVCM may reveal increased subepithelial reflectivity, activated keratocytes, and stromal scarring. The differential diagnosis of SNCD includes several ocular surface and corneal conditions that present with similar clinical features, particularly subepithelial corneal opacities or nodules. The presence of Salzmann-like nodules observed in interstitial keratitis is an important differential diagnosis, where chronic inflammation can lead to subepithelial fibrosis and nodule formation, mimicking SNCD. Band keratopathy, characterized by calcium deposition in the superficial cornea, may also present with gray-white opacities. However, these typically follow a horizontal interpalpebral distribution and have a distinct crystalline appearance. Epithelial basement membrane dystrophy, particularly when presenting with elevated map-dot-fingerprint patterns, can be mistaken for Salzmann nodules due to the irregularities on the anterior corneal surface. Pingueculae that extend onto the cornea can cause localized elevation and opacity, leading to diagnostic confusion, especially in the nasal interpalpebral region. Lastly, peripheral hypertrophic subepithelial corneal degeneration (PHSCD), a condition with peripheral subepithelial fibrosis and opacification, should be considered, particularly when nodules are located in the peripheral cornea. Accurate clinical assessment, aided by slit-lamp biomicroscopy and anterior segment imaging, is crucial for distinguishing these entities and ensuring appropriate management. SNCD is a slowly progressive condition, but it may remain stable for many years. Spontaneous regression is rare. Some patients may experience episodic symptom exacerbation due to concurrent ocular surface disease. Without treatment, nodules may enlarge and coalesce, contributing to more pronounced vision loss. However, many patients adapt well with minimal intervention. Symptom severity does not always correlate with nodule size. Even small lesions located centrally may significantly degrade vision due to irregular astigmatism or localized tear film disruption. Management strategies for SNCD are individualized and primarily depend on the severity of symptoms, the anatomical location of the nodules, and the presence of underlying or associated ocular surface disease. In mild cases, conservative treatment is often effective, typically involving regular use of lubricating eye drops to alleviate discomfort and maintain the stability of the tear film. Anti-inflammatory therapy, such as topical corticosteroids or cyclosporine, may be employed to control subclinical or overt inflammation. Additionally, addressing contributory conditions such as meibomian gland dysfunction or chronic blepharitis is critical in halting progression and preventing recurrence. In cases where conservative management fails or vision is significantly impaired due to central nodular encroachment or irregular astigmatism, surgical intervention becomes necessary. Superficial keratectomy involves the manual excision of nodules and is the first-line surgical approach in many patients. PTK, performed with an excimer laser, enables precise ablation and smoothing of the corneal surface, often yielding excellent visual outcomes. The adjunctive use of mitomycin-C may reduce recurrence by limiting fibroblastic activity. In advanced cases with deeper stromal involvement or a history of previous surgical failure, lamellar or penetrating keratoplasty may be required to restore visual function. Regardless of the chosen approach, long-term management of the ocular surface is crucial for minimizing the risk of recurrence and optimizing surgical outcomes. Postoperative recurrence is possible, especially in cases with ongoing ocular surface inflammation. Long-term lubrication and anti-inflammatory therapy may help reduce the risk of recurrence. Unlike infectious or neoplastic conditions, SNCD does not exhibit contiguous tissue spread. However, the disease can progress centripetally, especially in the presence of unaddressed risk factors. Nodules may increase in size or number, and new lesions may form in adjacent corneal areas. The prognosis for SNCD is generally favorable. Vision can often be preserved or restored with timely and appropriate treatment—most patients who undergo superficial keratectomy or PTK experience significant symptomatic and visual improvement. However, recurrence rates vary and depend largely on the adequacy of management of coexisting ocular surface disease. SNCD is a clinically significant yet underrecognized degenerative corneal disorder. Although indolent, its impact on visual function and quality of life can be profound, particularly when lesions encroach on the visual axis or induce irregular astigmatism. Early recognition, accurate diagnosis, and a tailored approach to management are essential. Understanding the anatomical basis, natural course, and associated risk factors is key for clinicians to provide evidence-based, interprofessional care aimed at preventing vision loss and minimizing recurrence.
A Rare Corneal Scenario: Concurrent Diagnosis of Epithelial Basement Membrane Dystrophy and Crocodile Shagreen.
Publicações recentes
Recurrence Rate After Phototherapeutic Keratectomy in Epithelial-Stromal TGFBI Corneal Dystrophies Compared With Epithelial Basement Membrane Dystrophy.
Superficial Keratectomy with Cryopreserved Amniotic Membrane for Ocular Surface Optimization in Patients with Epithelial Basement Membrane Dystrophy.
A Rare Corneal Scenario: Concurrent Diagnosis of Epithelial Basement Membrane Dystrophy and Crocodile Shagreen.
Meesmann Corneal Dystrophy with Epithelial Basement Membrane Abnormalities: Clinical and Genetic Analysis of Two Families with Novel and Known Mutations in KRT3 and KRT12.
Analysis of corneal wavefront aberrations and corneal densitometry in eyes with epithelial basement membrane dystrophy.
📚 EuropePMC51 artigos no totalmostrando 49
Superficial Keratectomy with Cryopreserved Amniotic Membrane for Ocular Surface Optimization in Patients with Epithelial Basement Membrane Dystrophy.
Clinical optometryA Rare Corneal Scenario: Concurrent Diagnosis of Epithelial Basement Membrane Dystrophy and Crocodile Shagreen.
Turkish journal of ophthalmologyMeesmann Corneal Dystrophy with Epithelial Basement Membrane Abnormalities: Clinical and Genetic Analysis of Two Families with Novel and Known Mutations in KRT3 and KRT12.
International journal of molecular sciencesAnalysis of corneal wavefront aberrations and corneal densitometry in eyes with epithelial basement membrane dystrophy.
International ophthalmologyHigher-Order Aberrations, Corneal Density, and Visual Acuity After Excimer Laser Phototherapeutic Keratectomy (PTK) for Epithelial Basement Membrane Dystrophy.
Journal of ophthalmologyA low-cost, easy-to-implement optical add-on to convert a retinal OCT into a ultra-high-resolution corneal OCT.
Zeitschrift fur medizinische PhysikConcurrent Meesmann Corneal Dystrophy and Epithelial Basement Membrane Dystrophy.
OphthalmologyEnhancing Visual Quality: The Impact of Alcohol-Assisted Delamination on Corneal Aberrations in Patients with Central Epithelial Basement Membrane Dystrophy.
Journal of clinical medicineAblation Depth-Dependent Survival Analysis of Phototherapeutic Keratectomy for Recurrent Corneal Erosion Syndrome.
Ophthalmology and therapyCorneal Epithelial Wavefront Error as a Novel Diagnostic Marker for Epithelial Basement Membrane Dystrophy.
Life (Basel, Switzerland)The Co-Occurrence of 22q11.2 Deletion Syndrome and Epithelial Basement Membrane Dystrophy: A Case Report and Review of the Literature.
Life (Basel, Switzerland)Early Detection of Subclinical Corneal Abnormalities: Biophotonic Imaging Reveals Hyporeflective Bleb-Like Structures in Asymptomatic Eyes.
The American journal of case reportsPilot Safety Study of a Microfabricated Device for Anterior Stromal Puncture to Treat Corneal Epithelial Disease in the Optical Axis.
CorneaClinical outcomes of epithelial basement membrane dystrophy after keratorefractive lenticule extraction.
Journal of cataract and refractive surgeryMatrix Metalloproteinases and the Pathogenesis of Recurrent Corneal Erosions and Epithelial Basement Membrane Dystrophy.
BiologyAcuity and Quality of Vision in Eyes with Epithelial Basement Membrane Dystrophy after Regular Pseudophakia.
Journal of clinical medicineAnterior Segment Swept Source Optical Coherence Tomography and In Vivo Confocal Microscopy Findings in a Case With Bleb-Like Epithelial Basal Membrane Dystrophy.
CorneaOcular surface optimization before cataract surgery.
Saudi journal of ophthalmology : official journal of the Saudi Ophthalmological Society[Preliminary Examination Before Cataract Surgery].
Klinische Monatsblatter fur Augenheilkunde[Phototherapeutic keratectomy for recurrent corneal erosion of various epithelial origins: influence of depth of ablation on pachymetry and refraction].
Die OphthalmologieCorneal Epithelial Thickness Mapping in the Diagnosis of Ocular Surface Disorders Involving the Corneal Epithelium: A Comparative Study.
CorneaTreatment of Epithelial Basement Membrane Dystrophy to Optimize the Ocular Surface Prior to Cataract Surgery.
Clinical ophthalmology (Auckland, N.Z.)[Phototherapeutic keratectomy for epithelial basement membrane dystrophy : Impact of excimer laser systems used on the changes of visual acuity, refraction and astigmatism].
Die OphthalmologiePhototherapeutic keratectomy versus epithelial debridement combined with anterior stromal puncture or diamond burr for recurrent corneal erosions.
Canadian journal of ophthalmology. Journal canadien d'ophtalmologieOptimizing the ocular surface prior to cataract surgery.
Current opinion in ophthalmologyIn vivo confocal microscopic images of atypical amiodarone-induced keratopathy in patient with epithelial basement membrane dystrophy.
American journal of ophthalmology case reportsAssessment of corneal epithelial thickness mapping in epithelial basement membrane dystrophy.
PloS oneMatching the Patient to the Intraocular Lens: Preoperative Considerations to Optimize Surgical Outcomes.
OphthalmologyEpithelial basement membrane dystrophy after femtosecond laser-assisted LASIK successfully treated with in vivo confocal microscopy-assisted photorefractive keratectomy.
Journal of cataract and refractive surgeryEfficacy of a Topical Heparan Sulfate Mimetic Polymer on Ocular Surface Discomfort in Patients with Cogan's Epithelial Basement Membrane Dystrophy.
Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and TherapeuticsImpact of epithelial basement membrane dystrophy and Salzmann nodular degeneration on biometry measurements.
Journal of cataract and refractive surgeryFalse corneal ectasia in patients referred for corneal crosslinking, topography-guided photorefractive keratectomy, and intrastromal corneal rings.
Canadian journal of ophthalmology. Journal canadien d'ophtalmologieA CARE-compliant article: optical coherence tomography for epithelial basement membrane dystrophy: A case report.
MedicineRefractive surprise after routine cataract surgery with multifocal IOLs attributable to corneal epithelial basement membrane dystrophy.
Journal of cataract and refractive surgeryRecurrent corneal erosion: a comprehensive review.
Clinical ophthalmology (Auckland, N.Z.)[Epithelial Dystrophies of the Cornea].
Klinische Monatsblatter fur Augenheilkunde[Corneal dystrophies in optical coherence tomography].
Der Ophthalmologe : Zeitschrift der Deutschen Ophthalmologischen GesellschaftSuperficial keratectomy: indications and outcomes.
Canadian journal of ophthalmology. Journal canadien d'ophtalmologiePrevalence of ocular surface dysfunction in patients presenting for cataract surgery evaluation.
Journal of cataract and refractive surgery[Recurrent Corneal Erosions in Epithelial Corneal Dystrophies].
Klinische Monatsblatter fur AugenheilkundeEpithelial basement membrane dystrophy after femtosecond laser-assisted laser in situ keratomileusis.
Canadian journal of ophthalmology. Journal canadien d'ophtalmologiePhototherapeutic keratectomy for epithelial basement membrane dystrophy.
Clinical ophthalmology (Auckland, N.Z.)Genotype-Phenotype Correlation for TGFBI Corneal Dystrophies Identifies p.(G623D) as a Novel Cause of Epithelial Basement Membrane Dystrophy.
Investigative ophthalmology & visual scienceUncovering the profile of mutations of transforming growth factor beta-induced gene in Chinese corneal dystrophy patients.
International journal of ophthalmology[Epithelial basement membrane dystrophy in a patient with keratoconus].
Journal francais d'ophtalmologieComparative Study of Anterior Eye Segment Measurements with Spectral Swept-Source and Time-Domain Optical Coherence Tomography in Eyes with Corneal Dystrophies.
BioMed research internationalLong-Term Outcomes of Epithelial Debridement and Diamond Burr Polishing for Corneal Epithelial Irregularity and Recurrent Corneal Erosion.
Cornea[Imaging of corneal dystrophies: Correlations between en face anterior segment OCT and in vivo confocal microscopy].
Journal francais d'ophtalmologieFourier-domain optical coherence tomography imaging in corneal epithelial basement membrane dystrophy: a structural analysis.
American journal of ophthalmologyAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Distrofia corneana, microcística.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Distrofia corneana, microcística
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Meesmann Corneal Dystrophy with Epithelial Basement Membrane Abnormalities: Clinical and Genetic Analysis of Two Families with Novel and Known Mutations in KRT3 and KRT12.
- Superficial Keratectomy with Cryopreserved Amniotic Membrane for Ocular Surface Optimization in Patients with Epithelial Basement Membrane Dystrophy.
- Analysis of corneal wavefront aberrations and corneal densitometry in eyes with epithelial basement membrane dystrophy.
- Concurrent Meesmann Corneal Dystrophy and Epithelial Basement Membrane Dystrophy.
- A Rare Corneal Scenario: Concurrent Diagnosis of Epithelial Basement Membrane Dystrophy and Crocodile Shagreen.
- Recurrence Rate After Phototherapeutic Keratectomy in Epithelial-Stromal TGFBI Corneal Dystrophies Compared With Epithelial Basement Membrane Dystrophy.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:98956(Orphanet)
- OMIM OMIM:121820(OMIM)
- MONDO:0007375(MONDO)
- GARD:9732(GARD (NIH))
- Variantes catalogadas(ClinVar)
- Busca completa no PubMed(PubMed)
- Q4162388(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar