Perda auditiva mista é caracterizada pela ocorrência simultânea de uma desordem do sistema auditivo com dois componentes: um componente condutivo e um componente sensorial.
Introdução
O que você precisa saber de cara
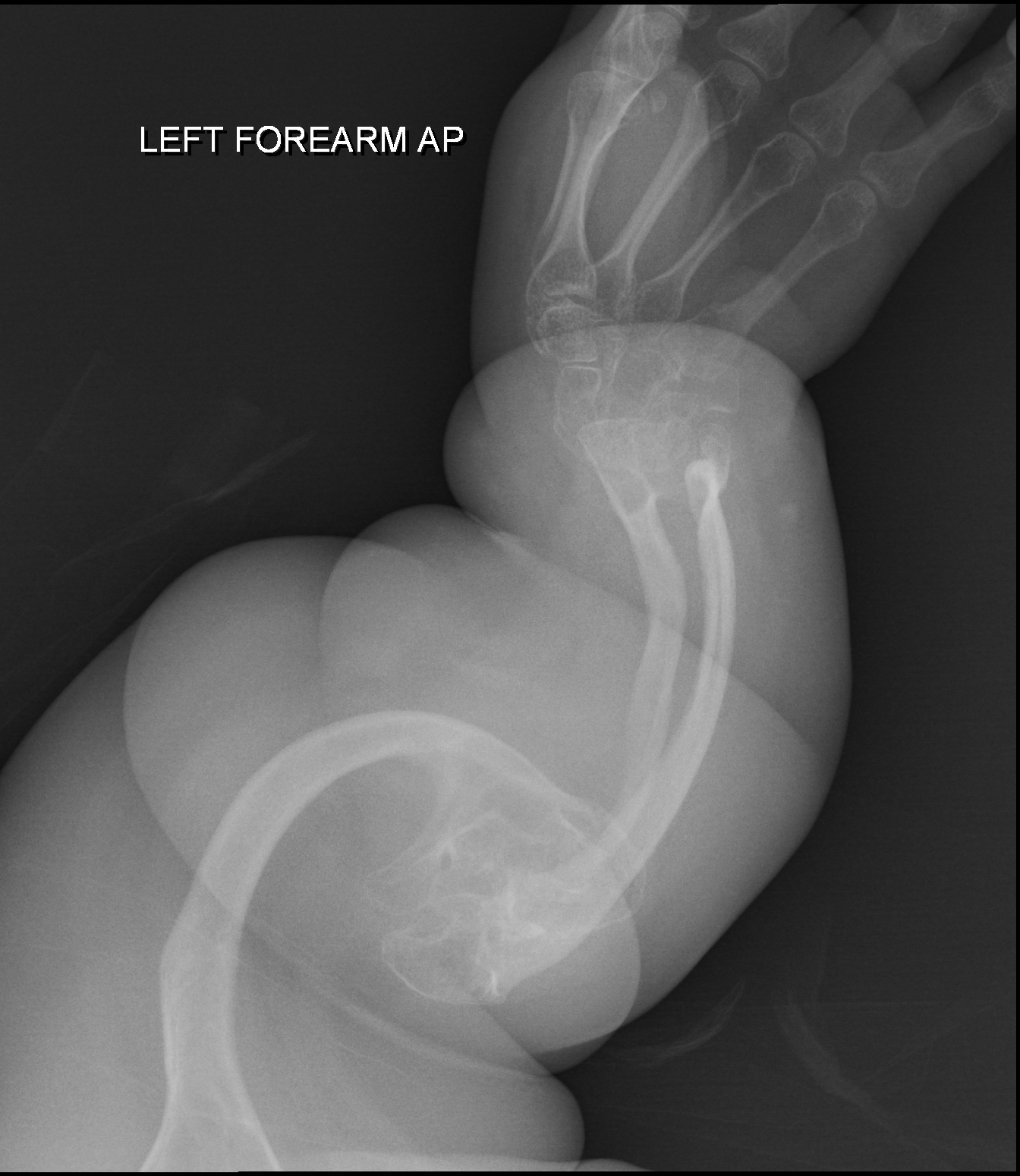
Osteogenesis imperfecta com massa óssea elevada é uma doença autossômica dominante rara caracterizada por densidade mineral óssea aumentada. Mutações nos genes COL1A2, COL1A1 ou BMP1 estão associadas a este fenótipo.
Escala de raridade
<1/50kMuito rara
1/20kRara
1/10kPouco freq.
1/5kIncomum
1/2k
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 1 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Genes associados
3 genes identificados com associação a esta condição. Padrão de herança: Autosomal dominant.
Type I collagen is a member of group I collagen (fibrillar forming collagen)
Secreted, extracellular space, extracellular matrix
Ehlers-Danlos syndrome, arthrochalasia type, 2
A form of Ehlers-Danlos syndrome, a connective tissue disorder characterized by hyperextensible skin, atrophic cutaneous scars due to tissue fragility and joint hyperlaxity. EDSARTH2 is an autosomal dominant condition characterized by frequent congenital hip dislocation and extreme joint laxity with recurrent joint subluxations and minimal skin involvement.
Type I collagen is a member of group I collagen (fibrillar forming collagen)
Secreted, extracellular space, extracellular matrix
Caffey disease
An autosomal dominant disorder characterized by an infantile episode of massive subperiosteal new bone formation that typically involves the diaphyses of the long bones, mandible, and clavicles. The involved bones may also appear inflamed, with painful swelling and systemic fever often accompanying the illness. The bone changes usually begin before 5 months of age and resolve before 2 years of age.
Metalloprotease that plays key roles in regulating the formation of the extracellular matrix (ECM) via processing of various precursor proteins into mature functional enzymes or structural proteins (PubMed:33206546). Thereby participates in several developmental and physiological processes such as cartilage and bone formation, muscle growth and homeostasis, wound healing and tissue repair (PubMed:32636307, PubMed:33169406). Roles in ECM formation include cleavage of the C-terminal propeptides fr
Golgi apparatus, trans-Golgi networkSecreted, extracellular space, extracellular matrixSecreted
Osteogenesis imperfecta 13
An autosomal recessive form of osteogenesis imperfecta, a disorder of bone formation characterized by low bone mass, bone fragility and susceptibility to fractures after minimal trauma. Disease severity ranges from very mild forms without fractures to intrauterine fractures and perinatal lethality. Extraskeletal manifestations, which affect a variable number of patients, are dentinogenesis imperfecta, hearing loss, and blue sclerae. OI13 is characterized by normal teeth, faint blue sclerae, severe growth deficiency, severe bone deformity, and recurrent fractures affecting both upper and lower limbs.
Variantes genéticas (ClinVar)
2,942 variantes patogênicas registradas no ClinVar.
Vias biológicas (Reactome)
30 vias biológicas associadas aos genes desta condição.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Osteogenesis imperfecta com massa óssea elevada
Centros de Referência SUS
24 centros habilitados pelo SUS para Osteogenesis imperfecta com massa óssea elevada
Centros para Osteogenesis imperfecta com massa óssea elevada
Detalhes dos centros
Hospital Universitário Prof. Edgard Santos (HUPES)
R. Dr. Augusto Viana, s/n - Canela, Salvador - BA, 40110-060 · CNES 0003808
Serviço de Referência
Hospital Infantil Albert Sabin
R. Tertuliano Sales, 544 - Vila União, Fortaleza - CE, 60410-794 · CNES 2407876
Serviço de Referência
Hospital de Apoio de Brasília (HAB)
AENW 3 Lote A Setor Noroeste - Plano Piloto, Brasília - DF, 70684-831 · CNES 0010456
Serviço de Referência
Hospital Estadual Infantil e Maternidade Alzir Bernardino Alves (HIABA)
Av. Min. Salgado Filho, 918 - Soteco, Vila Velha - ES, 29106-010 · CNES 6631207
Serviço de Referência
Hospital das Clínicas da UFG
Rua 235 QD. 68 Lote Área, Nº 285, s/nº - Setor Leste Universitário, Goiânia - GO, 74605-050 · CNES 2338424
Serviço de Referência
Hospital Universitário da UFJF
R. Catulo Breviglieri, Bairro - s/n - Santa Catarina, Juiz de Fora - MG, 36036-110 · CNES 2297442
Atenção Especializada
Hospital das Clínicas da UFMG
Av. Prof. Alfredo Balena, 110 - Santa Efigênia, Belo Horizonte - MG, 30130-100 · CNES 2280167
Serviço de Referência
Hospital Universitário Julio Müller (HUJM)
R. Luis Philippe Pereira Leite, s/n - Alvorada, Cuiabá - MT, 78048-902 · CNES 2726092
Atenção Especializada
Hospital Universitário João de Barros Barreto
R. dos Mundurucus, 4487 - Guamá, Belém - PA, 66073-000 · CNES 2337878
Serviço de Referência
Hospital Universitário Lauro Wanderley (HULW)
R. Tabeliao Estanislau Eloy, 585 - Castelo Branco, João Pessoa - PB, 58050-585 · CNES 0002470
Atenção Especializada
Instituto de Medicina Integral Prof. Fernando Figueira (IMIP)
R. dos Coelhos, 300 - Boa Vista, Recife - PE, 50070-902 · CNES 0000647
Serviço de Referência
Hospital Pequeno Príncipe
R. Des. Motta, 1070 - Água Verde, Curitiba - PR, 80250-060 · CNES 3143805
Serviço de Referência
Hospital Universitário Regional de Maringá (HUM)
Av. Mandacaru, 1590 - Parque das Laranjeiras, Maringá - PR, 87083-240 · CNES 2216108
Atenção Especializada
Hospital de Clínicas da UFPR
R. Gen. Carneiro, 181 - Alto da Glória, Curitiba - PR, 80060-900 · CNES 2364980
Serviço de Referência
Hospital Universitário Pedro Ernesto (HUPE-UERJ)
Blvd. 28 de Setembro, 77 - Vila Isabel, Rio de Janeiro - RJ, 20551-030 · CNES 2280221
Serviço de Referência
Instituto Nacional de Saúde da Mulher, da Criança e do Adolescente Fernandes Figueira (IFF/Fiocruz)
Av. Rui Barbosa, 716 - Flamengo, Rio de Janeiro - RJ, 22250-020 · CNES 2269988
Serviço de Referência
Hospital São Lucas da PUCRS
Av. Ipiranga, 6690 - Jardim Botânico, Porto Alegre - RS, 90610-000 · CNES 2232928
Serviço de Referência
Hospital de Clínicas de Porto Alegre (HCPA)
Rua Ramiro Barcelos, 2350 Bloco A - Av. Protásio Alves, 211 - Bloco B e C - Santa Cecília, Porto Alegre - RS, 90035-903 · CNES 2237601
Serviço de Referência
Hospital Universitário da UFSC (HU-UFSC)
R. Profa. Maria Flora Pausewang - Trindade, Florianópolis - SC, 88036-800 · CNES 2560356
Serviço de Referência
Hospital das Clínicas da FMUSP
R. Dr. Ovídio Pires de Campos, 225 - Cerqueira César, São Paulo - SP, 05403-010 · CNES 2077485
Serviço de Referência
Hospital de Base de São José do Rio Preto
Av. Brg. Faria Lima, 5544 - Vila Sao Jose, São José do Rio Preto - SP, 15090-000 · CNES 2079798
Atenção Especializada
Hospital de Clínicas da UNICAMP
R. Vital Brasil, 251 - Cidade Universitária, Campinas - SP, 13083-888 · CNES 2748223
Serviço de Referência
Hospital de Clínicas de Ribeirão Preto (HCRP-USP)
R. Ten. Catão Roxo, 3900 - Vila Monte Alegre, Ribeirão Preto - SP, 14015-010 · CNES 2082187
Serviço de Referência
UNIFESP / Hospital São Paulo
R. Napoleão de Barros, 715 - Vila Clementino, São Paulo - SP, 04024-002 · CNES 2688689
Serviço de Referência
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Ensaios em destaque
Pesquisa e ensaios clínicos
0 ensaios clínicos encontrados.
Publicações mais relevantes
Posterior spinal fusion with pedicle screw-based constructs in osteogenesis imperfecta: a systematic review of surgical and radiographic outcomes.
Osteogenesis imperfecta (OI) is a rare genetic disorder characterized by bone fragility and severe spinal deformities, with scoliosis affecting up to 80% of patients and often progressing despite bracing. Surgical management is challenging due to poor bone quality and high complication risk. Advances in pedicle screw-based constructs and multimodal strategies, including traction and bisphosphonates, have improved outcomes and enabled the successful correction of deformities. This review analyzes radiographic and surgical results of modern posterior spinal fusion (PSF) in OI-associated spinal deformity. A systematic search of PubMed, Scopus, Embase, Cochrane Library, and Google Scholar (inception to May 2025) was performed using search terms such as "osteogenesis imperfecta", "brittle bone disease", "posterior spinal fusion", "spinal arthrodesis", "scoliosis" and "spinal deformity". Extracted data covered demographics, OI type, traction techniques, instrumentation, radiographic and surgical outcomes, complications, and patient-reported outcome measures (PROMs). The risk of bias was assessed using the MINORS tool, and reporting followed PRISMA guidelines. The initial search identified 264 articles, of which 8 met the inclusion criteria, including 149 patients with OI (mean age 15.5 years). All studies were retrospective case series (level IV evidence). Cement augmentation was used in 31.5% of cases and apical osteotomies in 37.5%. Preoperative main curves ranged from 75.5° to 96°, with a mean correction rate of 49.5% after PSF. Both coronal and sagittal radiographic parameters improved postoperatively. The mean operative time was 410.6 minutes, blood loss averaged 1,375 mL, and hospital stay was 7.9 days. The overall complication rate was 27.5%, with 10.7% requiring unplanned reoperation. Modern pedicle screw-based constructs appear to provide more consistent radiographic correction in OI-associated scoliosis compared to earlier in situ fusion techniques. Although these procedures still entail significant blood loss and long operative times, their complication rates remain acceptable given patient complexity. Future multicenter high-quality studies should focus on optimizing implant density, screw augmentation, rod material, osteotomies, and integrating navigation and new biomaterials to standardize treatment strategies.
Matrix-directed therapy losartan to identify the effect on the bone resorption marker carboxy-terminal crosslink of type I collagen telopeptide (CTX) in older adolescents and adults with osteogenesis imperfecta recruited from secondary care sites: the 'MOI-A' study; a randomised, phase 2/pilot, dose-escalating trial.
Osteogenesis imperfecta (OI) is the most common inherited cause of bone fragility (approximately 1 in 16 000). People with OI suffer bone fragility causing fractures, pain and deformity; sarcopenia causing fatigue and poor endurance; aortic root dilatation and hearing loss. No drug currently has market authorisation to treat OI in Europe. Current standard-of-care is multidisciplinary, with pharmacological interventions-primarily bisphosphonates-directed at increasing bone mass; however, such interventions are of equivocal efficacy. The structural damage that can accumulate as a result of repeated fractures over time may not be reversible. The lack of a treatment with clearly defined efficacy in terms of reducing fracture frequency or the sarcopenia, that is increasingly recognised in this condition, leads to the consideration of alternatives based on what is known about the molecular pathophysiology of the condition. For reasons that are currently unclear, transforming growth factor beta (TGFβ) pathway signalling is increased in OI, and both studies in mouse models and more recently also in humans suggest that reducing TGFβ pathway signalling could be of benefit in OI. This demonstrator project tests the hypothesis that losartan, an antihypertensive agent known to reduce circulating TGFβ, will reduce bone turnover and bone loss and have a positive effect on muscle function and quality of life in adults and older adolescents with OI. This is a phase 2/pilot, open-label, dose-escalating study. This study aims to identify the effective dose for losartan in this population to inform the design of a pivotal phase III study. The study aims to recruit 30 adolescents and adults aged 16 years and above with OI across secondary care study sites in the UK and Italy. Participants will be recruited from the patient populations attending for treatment of OI at the participating hospital sites or referred by clinicians at the Participant Identification Centres (PIC sites). Participants will be randomised to one of three 'final doses'-25, 50 or 75 mg losartan once daily. All participants will start on 25 mg once daily. Those assigned to higher 'final doses' will increase in 25 mg once daily increments on day 8 and day 15 following safety assessments. The primary outcome measures are to establish the effective dose of losartan in OI patients, based on maximal reduction in the bone resorption marker carboxy-terminal crosslink of type I collagen telopeptide (CTX) over the 24-week period of the study.Secondary outcome measures are to determine the changes in proxy efficacy outcomes for bone (turnover, mass, architecture and strength) using blood tests, high-resolution peripheral quantitative CT (HRpQCT), dual-energy X-ray absorptiometry (DXA) and muscle (strength) using the 'Timed Up and Go' test. In addition, the changes in quality of life, including pain and fatigue, will be evaluated by using a disease-specific tool (OI-QOL) and a validated generic tool (EQ-5D-5L-VAS). In the UK, the study protocol and amendments have been approved by the London Bridge Research Ethics Committee (REC reference: 23/LO/015) and by the Medicines and Healthcare products Regulatory Agency (MHRA). In Italy, the study protocol and amendments have been approved by the Italian and European ethics and regulatory authorities (Clinical Trials Information System European Union (CTIS EU) portal according to EU Regulation 536/2014). Final version of study protocol: Version 3.2, 05.03.2025. Final results will be disseminated in peer-reviewed journals through local OI, orthopaedic and other relevant clinical networks and at national and international meetings. Sheffield Children's National Health Service Foundation Trust (UK) and Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS) Istituto Ortopedico Rizzoli (Italy) are the joint study sponsors. ISRCTN (ISRCTN13317811).
Molecular and Clinical Landscape of Osteogenesis Imperfecta: Unraveling Autosomal Recessive Forms, Therapeutic Outcomes, and Bone Mineral Density in Carriers.
Osteogenesis imperfecta (OI) is a heritable disorder characterized by bone fragility and marked genetic and phenotypic heterogeneity. This study explores the molecular and clinical spectrum of OI, with a focus on autosomal recessive (AR) forms, therapeutic outcomes, and bone mineral density (BMD) in carriers of AR OI-associated gene variants from the Indian population. A total of 78 clinically suspected OI patients were analyzed, yielding a high diagnostic rate of 92.3%. Exome sequencing was performed in all cases, with whole-genome sequencing in selected exome-negative cases. Autosomal dominant (AD) and AR OI accounted for 66% and 34% of cases, respectively. P3H1 (n = 11) was the most frequently implicated AR gene causing OI, followed by SERPINF1 (n = 5) and WNT1 (n = 4), with 79% of AR variants being novel. Phenotypic evaluation (n = 67) revealed fractures, short stature (87%), and bony deformities (84%) as predominant features. A rare homozygous COL1A1 variant was identified in one patient, while another patient harbored additional variants in AD OI genes, suggesting a potential digenic or modifier effect. Phenotypic severity followed the order from most to least severe: AR genes > COL1A2 (substitution and non-substitution) > COL1A1 (substitution > non-substitution). A self-designed, preliminary clinical severity scoring system ranked CRTAP followed by P3H1, as the AR genes associated with the most severe phenotypes. Therapeutic assessment showed a significant reduction in fracture incidence following zoledronate therapy only in the COL1A1 group, with no notable improvements in the COL1A2 or AR groups. Additionally, BMD evaluation in carrier parents of AR gene causing OI indicated a higher predisposition to low BMD among WNT1 gene carriers. However, these findings are preliminary and limited by small sample size. This study provides an extensive genotypic and phenotypic characterization of OI in the Indian population, with a focus on AR OI. It documents differential therapeutic responses among genetic subgroups and provides preliminary observations on BMD in carrier parents of AR OI-an aspect that has been less explored previously and suggest the need for tailored management strategies. The findings in this study also raise the possibility of genetic modifiers contributing to phenotypic variability, warranting further investigation.
Evaluation of the benefits of adapted physical activity in children and adolescents with osteogenesis imperfecta: the MOVE-OI trial.
Osteogenesis Imperfecta (OI) is a rare genetic disorder characterized by bone fragility and susceptibility to fractures. No curative treatment currently exists, and limited data are available on the effects of adapted physical activity (APA). This study evaluates the impact of APA on bone health, physical function, respiratory function, and quality of life in pediatric children with OI. The MOVE-OI trial (NCT04119388) is a prospective single-center study assessing the impact of a 12-month individualized APA program. Inclusion criteria included confirmed OI pathogenic variant, ages 6-18 years. Baseline (M0) and end-point (M12) assessments included clinical, radiological, and respiratory evaluations. The primary outcome was an improvement in the 6-min walk test (6MWT) distance. A non-parametric paired-test was performed for analysis. Thirty participants (16 males, median age 10.5 years) completed the program. A 17% increase in the 6MWT distance (p = 0.0007) was observed, with an average improvement of 98 m. No significant bone density or respiratory function changes were detected. Fracture incidence decreased (from 40 to 20%), and quality-of-life improvements were noted in participants with high baseline difficulty scores. APA improves endurance and physical capacity in children with OI. Multidisciplinary care and further research are needed to enhance long-term outcomes.
Viridicatol from the Deep-Sea-Derived Fungus Alleviates Bone Loss by Targeting the Wnt/SHN3 Pathway.
As an enticing bone anabolic target, short-term inhibition of Schnurri-3 (SHN3) resulted in high-bone mass due to augmented osteoblast activity. However, no studies are conducted to identify natural products targeting SHN3 inhibition. Herein, a screening strategy for the discovery of marine compounds that facilitate osteoblast differentiation by targeting SHN3 silencing is presented. One leading quinolinone alkaloid, viridicatol (VDC), isolated from deep-sea-derived fungus, vigorously promotes osteogenic differentiation via the Wnt/SHN3 signaling pathway in osteoblasts, thereby preventing osteoporosis while enhancing bone-fracture healing in a mouse model. Subsequently, the SDSSD (Ser, Asp, Ser, Ser, Asp) is further employed to engineer bone-targeting nanovesicles (BT-NVs) for the optimal delivery of VDC to osteoblasts, which mitigates the bone loss observed in a severe osteogenesis imperfecta model. Hence, these results initially uncover a promising marine natural product, VDC, targeting the Wnt/SHN3 pathway for the treatment of bone loss and highlighting its translational potential in clinical applications.
Publicações recentes
Matrix-directed therapy losartan to identify the effect on the bone resorption marker carboxy-terminal crosslink of type I collagen telopeptide (CTX) in older adolescents and adults with osteogenesis imperfecta recruited from secondary care sites: the 'MOI-A' study; a randomised, phase 2/pilot, dose-escalating trial.
Osteoclast-independent osteocyte dendrite defects in mice bearing the osteogenesis imperfecta-causing Sp7 R342C mutation.
Changes in lean mass and fat mass in children with Osteogenesis Imperfecta.
A Siglec-15 Antibody Promotes High Quality Bone Formation in Adult Female Mice With Osteogenesis Imperfecta.
Molecular and Clinical Landscape of Osteogenesis Imperfecta: Unraveling Autosomal Recessive Forms, Therapeutic Outcomes, and Bone Mineral Density in Carriers.
📚 EuropePMC3 artigos no totalmostrando 155
Posterior spinal fusion with pedicle screw-based constructs in osteogenesis imperfecta: a systematic review of surgical and radiographic outcomes.
European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research SocietyHarnessing Bone-Liver Crosstalk: A Dual-Action LYTAC Approach for Bone-Specific Accumulation and Liver-Specific Protein Degradation in Bone Disorders.
JACS AuCombined Treatment with a C-Type Natriuretic Peptide Analog and Bisphosphonate Enhances Bone Growth in Growing Mice with Osteogenesis Imperfecta: A Pilot Study.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchMatrix-directed therapy losartan to identify the effect on the bone resorption marker carboxy-terminal crosslink of type I collagen telopeptide (CTX) in older adolescents and adults with osteogenesis imperfecta recruited from secondary care sites: the 'MOI-A' study; a randomised, phase 2/pilot, dose-escalating trial.
BMJ openFabric-elasticity relationships of femoral head trabecular bone are similar in Type 2 diabetes and non-diabetic individuals.
Bone reportsMedication-Related Impacts on Pediatric Bone Health.
Journal of the Pediatric Orthopaedic Society of North AmericaBone microstructural and strength changes over one year in children with osteogenesis imperfecta are comparable to age- and sex-matched healthy controls.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchOsteoclast-independent osteocyte dendrite defects in mice bearing the osteogenesis imperfecta-causing Sp7 R342C mutation.
Bone researchChanges in lean mass and fat mass in children with Osteogenesis Imperfecta.
Journal of clinical densitometry : the official journal of the International Society for Clinical DensitometryA Siglec-15 Antibody Promotes High Quality Bone Formation in Adult Female Mice With Osteogenesis Imperfecta.
Journal of orthopaedic research : official publication of the Orthopaedic Research SocietyMolecular and Clinical Landscape of Osteogenesis Imperfecta: Unraveling Autosomal Recessive Forms, Therapeutic Outcomes, and Bone Mineral Density in Carriers.
Clinical geneticsAnti-transforming growth factor-β treatment shows increased bone mass and strength in a novel mouse model for osteogenesis imperfecta type I.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchSmall and porous ossicles, with flat stapes footplate and incudal fractures in the oim mouse model of osteogenesis imperfecta.
BoneSpontaneous Gallbladder Perforation in a Patient With Osteogenesis Imperfecta Type III: A Rare Case Report.
Annali italiani di chirurgiaEvaluation of the benefits of adapted physical activity in children and adolescents with osteogenesis imperfecta: the MOVE-OI trial.
Orphanet journal of rare diseasesViridicatol from the Deep-Sea-Derived Fungus Alleviates Bone Loss by Targeting the Wnt/SHN3 Pathway.
Advanced science (Weinheim, Baden-Wurttemberg, Germany)TBS as a complementary tool for assessing vertebral fractures and spinal deformity in children and adolescents with osteogenesis imperfecta.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USAEarly life functional transitions impact craniofacial morphology in osteogenesis imperfecta.
Anatomical record (Hoboken, N.J. : 2007)Non-invasive quantification of bone (re)modeling dynamics in adults with osteogenesis imperfecta treated with setrusumab using timelapse high-resolution peripheral-quantitative computed tomography.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchCrispant analysis in zebrafish as a tool for rapid functional screening of disease-causing genes for bone fragility.
eLifeOrthodontic Management in Pediatric Patients with Rare Diseases: Case Reports.
Journal of clinical medicineAcromial and scapular fractures after reverse shoulder arthroplasty: comparison of 3018 reverse total shoulders by inlay and onlay humeral component design.
Journal of shoulder and elbow surgeryGenetics and Bone Mineral Density Predict the Fractures in Adults With Osteogenesis Imperfecta: A Prospective Study.
The Journal of clinical endocrinology and metabolismBone Quality and Mineralization and Effects of Treatment in Osteogenesis Imperfecta.
Calcified tissue internationalMonoclonal antibody anti-sclerostin for treatment of pelvic insufficiency fractures in adult hypophosphatasia: A case report.
Trauma case reportsSetrusumab for the treatment of osteogenesis imperfecta: 12-month results from the phase 2b asteroid study.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchNutritional Behavior of Patients with Bone Diseases: A Cross-Sectional Study from Austria.
NutrientsCorrelation of serum DKK1 level with skeletal phenotype in children with osteogenesis imperfecta.
Journal of endocrinological investigationClinical features, treatment, and follow-up of OPPG and high-bone-mass disorders: LRP5 is a key regulator of bone mass.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USABone microarchitecture and strength assessment in adults with osteogenesis imperfecta using HR-pQCT: normative comparison and challenges.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchInfluence of zoledronic acid and pamidronate on tooth eruption in children with osteogenesis imperfecta.
BonePhysiological cell bioprinting density in human bone-derived cell-laden scaffolds enhances matrix mineralization rate and stiffness under dynamic loading.
Frontiers in bioengineering and biotechnologyImaging in osteogenesis imperfecta: Where we are and where we are going.
European journal of medical geneticsSafety and Efficacy of Denosumab in Children With Osteogenesis Imperfecta-the First Prospective Comparative Study.
The Journal of clinical endocrinology and metabolismEfficacy and Safety of Denosumab vs Zoledronic Acid in OI Adults: A Prospective, Open-Label, Randomized Study.
The Journal of clinical endocrinology and metabolismΤhe story of sclerostin inhibition: the past, the present, and the future.
Hormones (Athens, Greece)In Vivo Assessment of Skin Surface Pattern: Exploring Its Potential as an Indicator of Bone Biomechanical Properties.
Bioengineering (Basel, Switzerland)LRP5, Bone Mass Polymorphisms and Skeletal Disorders.
GenesOsteosclerotic Metaphyseal Dysplasia Due to a Likely Pathogenic LRRK1 Variant as a Cause of Recurrent Long Bone Fractures.
JBMR plusDenosumab use in osteogenesis imperfecta: an update on therapeutic approaches.
Annals of pediatric endocrinology & metabolismCorrelation of lipocalin 2 and glycolipid metabolism and body composition in a large cohort of children with osteogenesis imperfecta.
Journal of endocrinological investigationCyclic intravenous pamidronate for an infant with osteogenesis imperfecta type II.
BMJ case reportsPrevalence of Monogenic Bone Disorders in a Dutch Cohort of Atypical Femur Fracture Patients.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchDenosumab in pediatric bone disorders and the role of RANKL blockade: a narrative review.
Translational pediatricsCardiovascular abnormalities and its correlation with genotypes of children with osteogenesis imperfecta.
Frontiers in endocrinologySkeletal outcomes of patients with osteogenesis imperfecta during drug holiday of bisphosphonates: a real-world study.
Frontiers in endocrinologyOsteoporosis in children and young adults.
Best practice & research. Clinical rheumatologyBiomechanical, Microstructural and Material Properties of Tendon and Bone in the Young Oim Mice Model of Osteogenesis Imperfecta.
International journal of molecular sciencesThe role of osteocalcin in regulation of glycolipid metabolism and muscle function in children with osteogenesis imperfecta.
Frontiers in endocrinologyAlterations of bone material properties in growing Ifitm5/BRIL p.S42 knock-in mice, a new model for atypical type VI osteogenesis imperfecta.
BoneHigh Heterogeneity of Temporal Bone CT Aspects in Osteogenesis Imperfecta Is Not Linked to Hearing Loss.
Journal of clinical medicineDominant osteogenesis imperfecta with low bone turnover caused by a heterozygous SP7 variant.
BoneGenetic Diagnostics in Routine Osteological Assessment of Adult Low Bone Mass Disorders.
The Journal of clinical endocrinology and metabolism3D Image Registration Marginally Improves the Precision of HR-pQCT Measurements Compared to Cross-Sectional-Area Registration in Adults With Osteogenesis Imperfecta.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchA novel pathogenic variant at the C-terminal propeptide cleavage site of COL1A1, causing osteogenesis imperfecta with intrafamilial variability.
American journal of medical genetics. Part AData on body mass, glucose tolerance and bone phenotype of mice with osteogenesis imperfecta on long-term low-fat and high-fat diets.
Data in briefGrowth and Pubertal Features in a Cohort of 83 Patients with Osteogenesis Imperfecta.
Klinische PadiatrieTargeting TGF-β for treatment of osteogenesis imperfecta.
The Journal of clinical investigationThe recurrent homozygous translation start site variant in CCDC134 in an individual with severe osteogenesis imperfecta of non-Morrocan ancestry.
American journal of medical genetics. Part AX-Linked Osteogenesis Imperfecta Possibly Caused by a Novel Variant in PLS3.
GenesFabric-elasticity relationships of tibial trabecular bone are similar in osteogenesis imperfecta and healthy individuals.
BoneHigh bone mass phenotype in a cohort of patients with Osteogenesis Imperfecta caused due to BMP1 and C-propeptide cleavage variants in COL1A1.
Bone reportsOsteogenesis Imperfecta: Mechanisms and Signaling Pathways Connecting Classical and Rare OI Types.
Endocrine reviewsEnhancing osteogenesis of adipose-derived mesenchymal stem cells using gold nanostructure/peptide-nanopatterned graphene oxide.
Colloids and surfaces. B, BiointerfacesDentinogenesis Imperfecta and Caries in Osteogenesis Imperfecta among Vietnamese Children.
Dentistry journalAnalysis of bone architecture using fractal-based TX-Analyzer™ in adult patients with osteogenesis imperfecta.
BoneNovel homozygous variant in BMP1 associated with a rare osteogenesis imperfecta phenotype.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USAEvaluation of Body Composition in Paediatric Osteogenesis Imperfecta.
Journal of clinical densitometry : the official journal of the International Society for Clinical DensitometryIncreased cochlear otic capsule thickness and intracortical canal porosity in the oim mouse model of osteogenesis imperfecta.
Journal of structural biologyMultisite longitudinal calibration of HR-pQCT scanners and precision in osteogenesis imperfecta.
BoneIncidence and treatment of adult femoral fractures with osteogenesis imperfecta: An analysis of a center of 72 patients in Taiwan.
International journal of medical sciencesDo Bisphosphonates Alleviate Pain in Children? A Systematic Review.
Current osteoporosis reportsHigh Bone Mineral Density Osteogenesis Imperfecta in a Family with a Novel Pathogenic Variant in COL1A2.
Hormone research in paediatricsWhole-Exome Sequencing Identifies Novel Compound Heterozygous ZNF469 Mutations in Two Siblings with Mild Brittle Cornea Syndrome.
Calcified tissue internationalGenetic Burden Contributing to Extremely Low or High Bone Mineral Density in a Senior Male Population From the Osteoporotic Fractures in Men Study (MrOS).
JBMR plusFinite element analysis of bone strength in osteogenesis imperfecta.
BoneA Contemporary View of the Definition and Diagnosis of Osteoporosis in Children and Adolescents.
The Journal of clinical endocrinology and metabolismJuvenile osteoporosis with calvarial doughnuts: Progressive high-turnover bone loss responsive to bisphosphonate therapy.
Joint bone spineHybrid minigene splicing assay verifies the pathogenicity of a novel splice site variant in the COL1A1 gene of a chinese patient with osteogenesis imperfecta type I.
InjuryCombination therapy in the Col1a2G610C mouse model of Osteogenesis Imperfecta reveals an additive effect of enhancing LRP5 signaling and inhibiting TGFβ signaling on trabecular bone but not on cortical bone.
BoneHR-pQCT Measures of Bone Microarchitecture Predict Fracture: Systematic Review and Meta-Analysis.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchPretreatment with Pamidronate Decreases Bone Formation but Increases Callus Bone Volume in a Rat Closed Fracture Model.
Calcified tissue internationalIndividualized treatment with denosumab in children with osteogenesis imperfecta - follow up of a trial cohort.
Orphanet journal of rare diseasesSclerostin Antibody-Induced Changes in Bone Mass Are Site Specific in Developing Crania.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchComprehensive genetic analyses using targeted next-generation sequencing and genotype-phenotype correlations in 53 Japanese patients with osteogenesis imperfecta.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USASclerostin antibody reduces long bone fractures in the oim/oim model of osteogenesis imperfecta.
BoneNew Insights Into Monogenic Causes of Osteoporosis.
Frontiers in endocrinologyCOL1A2 p.Gly1066Val variant identified in a Han Chinese family with osteogenesis imperfecta type I.
Molecular genetics & genomic medicineOlecranon Fractures in Pediatric Patients With Osteogenesis Imperfecta.
Journal of pediatric orthopedicsGenotype-phenotype relationship in a large cohort of osteogenesis imperfecta patients with COL1A1 mutations revealed by a new scoring system.
Chinese medical journalImprovement of bone microarchitecture parameters after 12 months of treatment with asfotase alfa in adult patient with hypophosphatasia: Case report.
MedicinePhenotyping and genotyping of skeletal dysplasias: Evolution of a center and a decade of experience in India.
BoneNovel mutations in BMP1 induce a rare type of osteogenesis imperfecta.
Clinica chimica acta; international journal of clinical chemistryWnt1 is an Lrp5-independent bone-anabolic Wnt ligand.
Science translational medicineEffect of Anti-TGF-β Treatment in a Mouse Model of Severe Osteogenesis Imperfecta.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchTBS as a Tool to Differentiate the Impact of Antiresorptives onCortical and Trabecular Bone in Children With OsteogenesisImperfecta.
Journal of clinical densitometry : the official journal of the International Society for Clinical DensitometryInterventions for Improving Bone Mineral Density and Reducing Fracture Risk in Osteogenesis Imperfecta: A Mixed Treatment Comparison Network Meta-analysis of Randomized Controlled Clinical Trials.
Current clinical pharmacologyLoss of RANKL in osteocytes dramatically increases cancellous bone mass in the osteogenesis imperfecta mouse (oim).
Bone reportsComparison of Bone Microarchitecture Between Adult Osteogenesis Imperfecta and Early-Onset Osteoporosis.
Calcified tissue internationalMutations That Alter the Carboxy-Terminal-Propeptide Cleavage Site of the Chains of Type I Procollagen Are Associated With a Unique Osteogenesis Imperfecta Phenotype.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchElevated platelet counts in a cohort of children with moderate-severe osteogenesis imperfecta suggest that inflammation is present.
Archives of disease in childhoodNovel compound heterozygous mutations in SERPINH1 cause rare autosomal recessive osteogenesis imperfecta type X.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USAHypercalcemia and hypercalciuria during denosumab treatment in children with osteogenesis imperfecta type VI.
Journal of musculoskeletal & neuronal interactionsDiagnostic strategies and genotype-phenotype correlation in a large Indian cohort of osteogenesis imperfecta.
BoneDiagnosis, follow-up and management of sleep-disordered breathing in children with osteogenesis imperfecta.
Annals of physical and rehabilitation medicineNovel variant in Sp7/Osx associated with recessive osteogenesis imperfecta with bone fragility and hearing impairment.
BoneClassification of micro-CT images using 3D characterization of bone canal patterns in human osteogenesis imperfecta.
Proceedings of SPIE--the International Society for Optical EngineeringA novel COL1A2 C-propeptide cleavage site mutation causing high bone mass osteogenesis imperfecta with a regional distribution pattern.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USAApplication of next‑generation sequencing for molecular diagnosis in a large family with osteogenesis imperfecta type I.
Molecular medicine reportsHow frequent is osteogenesis imperfecta in patients with idiopathic osteoporosis?: Case reports.
MedicineGene mutation spectrum and genotype-phenotype correlation in a cohort of Chinese osteogenesis imperfecta patients revealed by targeted next generation sequencing.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USAIntraoperative bleeding in patients with osteogenesis imperfecta type III treated by Fassier-Duval femoral rodding: analysis of risk factors.
Journal of pediatric orthopedics. Part BSplenomegaly, myeloid lineage expansion and increased osteoclastogenesis in osteogenesis imperfecta murine.
BoneHypermineralization and High Osteocyte Lacunar Density in Osteogenesis Imperfecta Type V Bone Indicate Exuberant Primary Bone Formation.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchExploiting the WNT Signaling Pathway for Clinical Purposes.
Current osteoporosis reportsAn example of the utility of genomic analysis for fast and accurate clinical diagnosis of complex rare phenotypes.
Orphanet journal of rare diseasesSerum creatine kinase isoenzymes in children with osteogenesis imperfecta.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USAClinical characteristics and the identification of novel mutations of COL1A1 and COL1A2 in 61 Chinese patients with osteogenesis imperfecta.
Molecular medicine reportsSingle dose of bisphosphonate preserves gains in bone mass following cessation of sclerostin antibody in Brtl/+ osteogenesis imperfecta model.
BoneNovel mutations in FKBP10 in Chinese patients with osteogenesis imperfecta and their treatment with zoledronic acid.
Journal of human geneticsChallenges of Fracture Management for Adults With Osteogenesis Imperfecta.
OrthopedicsPhenotypic variability in patients with osteogenesis imperfecta caused by BMP1 mutations.
American journal of medical genetics. Part AA Novel ANO5 Mutation Causing Gnathodiaphyseal Dysplasia With High Bone Turnover Osteosclerosis.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchGenetics of osteoporosis: searching for candidate genes for bone fragility.
Archives of endocrinology and metabolismNext-generation sequencing of common osteogenesis imperfecta-related genes in clinical practice.
Scientific reportsEnhanced Wnt signaling improves bone mass and strength, but not brittleness, in the Col1a1(+/mov13) mouse model of type I Osteogenesis Imperfecta.
BoneDistinctive skeletal phenotype in high bone mass osteogenesis imperfecta due to a COL1A2 cleavage site mutation.
American journal of medical genetics. Part ASkeletal phenotypes in adult patients with osteogenesis imperfecta-correlations with COL1A1/COL1A2 genotype and collagen structure.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USAPamidronate treatment for osteogenesis imperfecta in black South Africans.
South African medical journal = Suid-Afrikaanse tydskrif vir geneeskundeBone mineral properties in growing Col1a2(+/G610C) mice, an animal model of osteogenesis imperfecta.
BoneThe genetics of bone mass and susceptibility to bone diseases.
Nature reviews. RheumatologyEffect of high-dose vitamin D supplementation on bone density in youth with osteogenesis imperfecta: A randomized controlled trial.
BoneTwo novel mutations in TMEM38B result in rare autosomal recessive osteogenesis imperfecta.
Journal of human geneticsAAV8-mediated expression of N-acetylglucosamine-1-phosphate transferase attenuates bone loss in a mouse model of mucolipidosis II.
Molecular genetics and metabolismImpaired bone remodeling in children with osteogenesis imperfecta treated and untreated with bisphosphonates: the role of DKK1, RANKL, and TNF-α.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USAGenetic control of bone mass.
Molecular and cellular endocrinologyDevelopmental Stage-dependent Regulation of Prolyl 3-Hydroxylation in Tendon Type I Collagen.
The Journal of biological chemistry[Pharmacological treatment of other types of secondary osteoporosis].
Nihon rinsho. Japanese journal of clinical medicineShort-Term Safety of Zoledronic Acid in Young Patients With Bone Disorders: An Extensive Institutional Experience.
The Journal of clinical endocrinology and metabolismPersistance and Compliance with Osteroporosis Therapies Among Women in a Commercially Insured Population in the United States.
Journal of managed care & specialty pharmacyBone mass and mineralization in osteogenesis imperfecta.
Wiener medizinische Wochenschrift (1946)Osteogenesis imperfecta type III and hypogonadotropic hypogonadism result in severe bone loss: a case report.
Wiener medizinische Wochenschrift (1946)Myostatin deficiency partially rescues the bone phenotype of osteogenesis imperfecta model mice.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USAIntravenous Bisphosphonate Therapy of Young Children With Osteogenesis Imperfecta: Skeletal Findings During Follow Up Throughout the Growing Years.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral ResearchComparison of DXA Scans and Conventional X-rays for Spine Morphometry and Bone Age Determination in Children.
Journal of clinical densitometry : the official journal of the International Society for Clinical DensitometryENDOCRINOLOGY AND ADOLESCENCE: Osteoporosis in children: diagnosis and management.
European journal of endocrinologyComplete Remodeling After Conservative Treatment of a Severely Angulated Odontoid Fracture in a Patient With Osteogenesis Imperfecta: A Case Report.
SpineWhole-exome sequencing identifies de novo mutation in the COL1A1 gene to underlie the severe osteogenesis imperfecta.
Human genomicsBone structure assessed by HR-pQCT, TBS and DXL in adult patients with different types of osteogenesis imperfecta.
Osteoporosis international : a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USASevere osteogenesis imperfecta Type-III and its challenging treatment in newborn and preschool children. A systematic review.
InjuryMicrostructure and compressive mechanical properties of cortical bone in children with osteogenesis imperfecta treated with bisphosphonates compared with healthy children.
Journal of the mechanical behavior of biomedical materialsPolymorphisms associated with low bone mass and high risk of atraumatic fracture.
Physiological researchDevelopment of a high-throughput resequencing array for the detection of pathogenic mutations in osteogenesis imperfecta.
PloS oneReliability of vertebral fractures assessment (VFA) in children with osteogenesis imperfecta.
Calcified tissue internationalUnique micro- and nano-scale mineralization pattern of human osteogenesis imperfecta type VI bone.
BoneAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Osteogenesis imperfecta com massa óssea elevada.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Osteogenesis imperfecta com massa óssea elevada
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Posterior spinal fusion with pedicle screw-based constructs in osteogenesis imperfecta: a systematic review of surgical and radiographic outcomes.European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society· 2026· PMID 41854906mais citado
- Matrix-directed therapy losartan to identify the effect on the bone resorption marker carboxy-terminal crosslink of type I collagen telopeptide (CTX) in older adolescents and adults with osteogenesis imperfecta recruited from secondary care sites: the 'MOI-A' study; a randomised, phase 2/pilot, dose-escalating trial.
- Molecular and Clinical Landscape of Osteogenesis Imperfecta: Unraveling Autosomal Recessive Forms, Therapeutic Outcomes, and Bone Mineral Density in Carriers.
- Evaluation of the benefits of adapted physical activity in children and adolescents with osteogenesis imperfecta: the MOVE-OI trial.
- Viridicatol from the Deep-Sea-Derived Fungus Alleviates Bone Loss by Targeting the Wnt/SHN3 Pathway.
- Osteoclast-independent osteocyte dendrite defects in mice bearing the osteogenesis imperfecta-causing Sp7 R342C mutation.
- Changes in lean mass and fat mass in children with Osteogenesis Imperfecta.
- A Siglec-15 Antibody Promotes High Quality Bone Formation in Adult Female Mice With Osteogenesis Imperfecta.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:314029(Orphanet)
- MONDO:0017791(MONDO)
- Osteogenese Imperfeita(PCDT · Ministério da Saúde)
- GARD:21366(GARD (NIH))
- Variantes catalogadas(ClinVar)
- Busca completa no PubMed(PubMed)
- Q55787374(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar