É um tipo de deficiência da enzima LCAT (lecithin-cholesterol acyltransferase) caracterizada clinicamente por opacidades na córnea, anemia hemolítica e insuficiência renal. Em exames de laboratório, ela se apresenta com colesterol HDL (o "colesterol bom") muito baixo e a falta completa da enzima LCAT.
Introdução
O que você precisa saber de cara
É um tipo de deficiência da enzima LCAT (lecithin-cholesterol acyltransferase) caracterizada clinicamente por opacidades na córnea, anemia hemolítica e insuficiência renal. Em exames de laboratório, ela se apresenta com colesterol HDL (o "colesterol bom") muito baixo e a falta completa da enzima LCAT.
Escala de raridade
<1/50kMuito rara
1/20kRara
1/10kPouco freq.
1/5kIncomum
1/2k
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Partes do corpo afetadas
+ 5 sintomas em outras categorias
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 11 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Genes associados
1 gene identificado com associação a esta condição. Padrão de herança: Autosomal recessive, Not applicable.
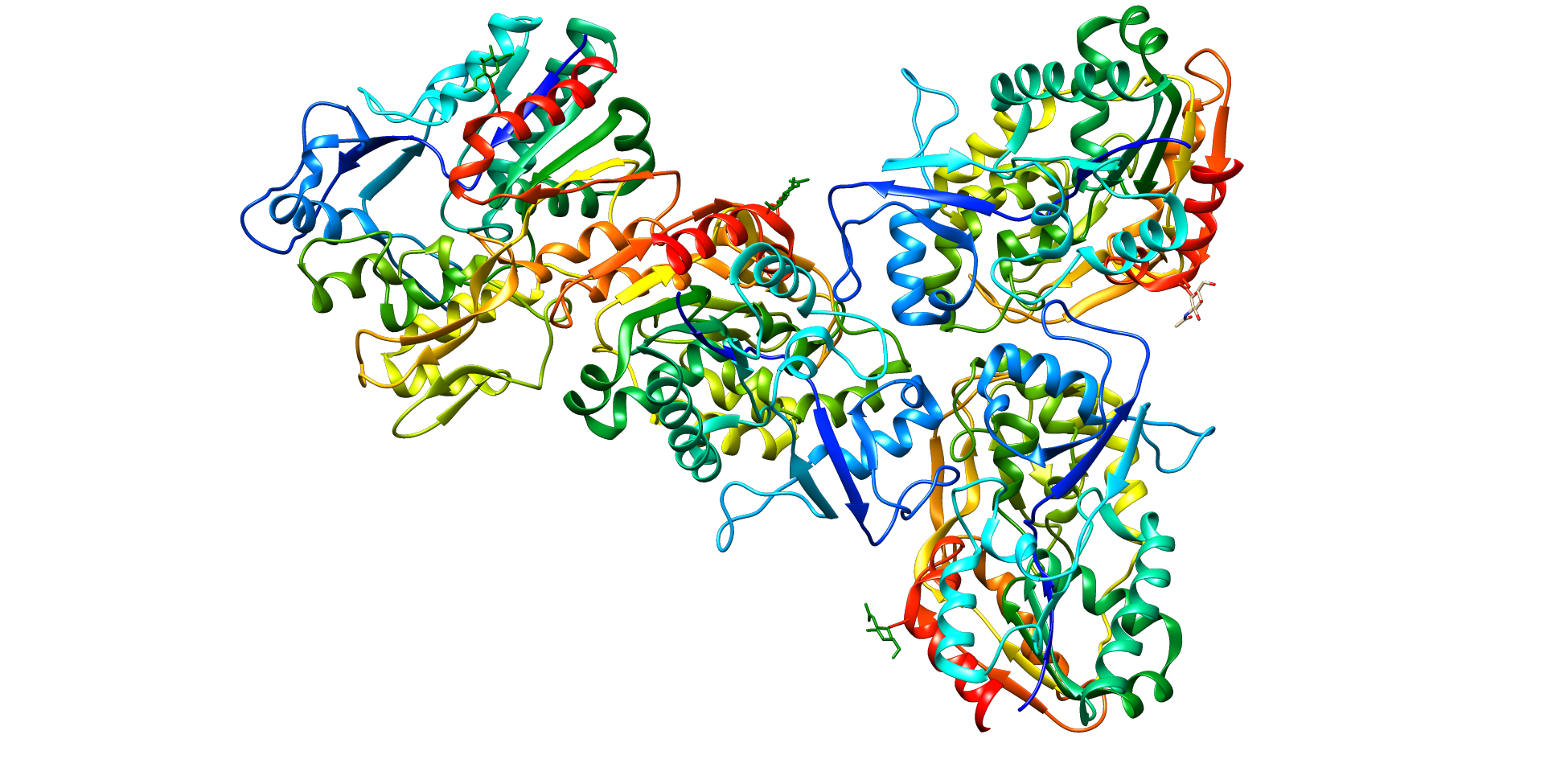
Central enzyme in the extracellular metabolism of plasma lipoproteins. Synthesized mainly in the liver and secreted into plasma where it converts cholesterol and phosphatidylcholines (lecithins) to cholesteryl esters and lysophosphatidylcholines on the surface of high and low density lipoproteins (HDLs and LDLs) (PubMed:10329423, PubMed:19065001, PubMed:26195816). The cholesterol ester is then transported back to the liver. Has a preference for plasma 16:0-18:2 or 18:O-18:2 phosphatidylcholines
Secreted
Lecithin-cholesterol acyltransferase deficiency
A disorder of lipoprotein metabolism characterized by inadequate esterification of plasmatic cholesterol. Two clinical forms are recognized: complete LCAT deficiency and fish-eye disease. LCATD is generally referred to the complete form which is associated with absence of both alpha and beta LCAT activities resulting in esterification anomalies involving both HDL (alpha-LCAT activity) and LDL (beta-LCAT activity). It causes a typical triad of diffuse corneal opacities, target cell hemolytic anemia, and proteinuria with renal failure.
Variantes genéticas (ClinVar)
77 variantes patogênicas registradas no ClinVar.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Deficiência de LCAT familiar
Selecione um estado ou use sua localização para ver resultados.
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Ensaios em destaque
🟢 Recrutando agora
1 pesquisa recrutando participantes. Converse com seu médico sobre a possibilidade de participar.
Outros ensaios clínicos
2 ensaios clínicos encontrados, 1 ativos.
Publicações mais relevantes
Lecithin-cholesterol acyltransferase deficiency with the finding of "zebra bodies": A diagnostic challenge in the context of suspected Fabry disease.
Lecithin-cholesterol acyltransferase (LCAT) deficiency is a rare autosomal recessive disorder resulting from mutations in the LCAT gene, which leads to abnormal lipoprotein metabolism. This results in markedly reduced high-density lipoprotein cholesterol and the accumulation of lipoprotein X, leading to renal, corneal, and hemolytic damage. Two clinical variants have been described: familial LCAT deficiency (FLD) and fish-eye disease (FED). We report the case of a 41-year-old male with a history of hypertension, tinnitus, and progressive hearing loss, who presented with bilateral corneal opacity since childhood. Laboratory studies revealed significant proteinuria (2.56 g/24 h), preserved renal function (creatinine 0.85 mg/dL), mild anemia (Hb 10.2 g/dL), and extremely low HDL cholesterol (1.3 mg/dL). Renal biopsy showed focal segmental glomerulosclerosis and mesangial expansion. Electron microscopy demonstrated concentric lamellar inclusions known as "zebra bodies," a finding typically associated with Fabry disease. However, α-galactosidase A activity was normal, and genetic testing for Fabry disease was negative. Further genetic analysis identified the variant c.757 p.(Gln253Argfs*11) in the LCAT gene, confirming the diagnosis of familial LCAT deficiency. This case highlights the importance of differentiating LCAT deficiency from Fabry disease, given their overlapping clinical and histological features. Moreover, it represents the first description of "zebra bodies" in LCAT deficiency, emphasizing the diagnostic complexity and the need for a multidisciplinary approach to ensure accurate diagnosis and appropriate management.
Familial LCAT Deficiency and Low HDL-C Levels: In silico Characterization of Two Rare LCAT Missense Mutations.
Mutations in the lecithin-cholesterol acyltransferase (LCAT) gene, which catalyzes the esterification of cholesterol, result in two types of autosomal recessive disorders: Familial LCAT deficiency (FLD) and Fish Eye Disease (FED). While both phenotypes are characterized by corneal opacities and different forms of dyslipidemia, such as low levels of high-density lipoprotein-cholesterol (HDL-C), FLD exhibits more severe clinical manifestations like splenomegaly, anemia, and renal failure. We describe the first clinically and genetically confirmed case of FLD in Colombia which corresponds to a 46-year-old woman with corneal opacity, hypothyroidism, and dyslipidemia, who does not have any manifestations of renal failure, with two pathogenic heterozygous missense variants in the LCAT gene: LCAT (NM_000229.2):c.803G>A (p.Arg268His) and LCAT (NM_000229.2):c.368G>C (p.Arg123Pro). In silico analysis of the mutations predicted the physicochemical properties of the mutated protein, causing instability and potentially decreased LCAT function. These compound mutations highlight the clinical heterogeneity of the phenotypes associated with LCAT gene mutations. Lecithin cholesterol acyltransferase (LCAT) deficiency is a rare inherited syndrome characterized by the partial or complete absence of LCAT enzyme activity. LCAT is an enzyme attached to both high-density lipoprotein (HDL) and low-density lipoprotein (LDL) particles and is responsible for the esterification of cholesterol, a crucial step in the metabolism of HDL particles. Deficiency of LCAT leads to impaired HDL metabolism, resulting in abnormal lipid profiles and predisposing to complications. There are 2 clinical variants of the disease. The first is familial LCAT deficiency that involves a complete deficiency of the enzyme. The second is Fish-eye disease characterized by a partial deficiency of the enzyme.
Rescue of Familial Lecithin:Cholesterol Acyltranferase Deficiency Mutations with an Allosteric Activator.
Lecithin:cholesterol acyltransferase (LCAT) deficiencies represent severe disorders characterized by aberrant cholesterol esterification in plasma, leading to life-threatening conditions. This study investigates the efficacy of Compound 2, a piperidinyl pyrazolopyridine allosteric activator that binds the membrane-binding domain of LCAT, in rescuing the activity of LCAT variants associated with disease. The variants K218N, N228K, and G230R, all located in the cap and lid domains of LCAT, demonstrated notable activity restoration in response to Compound 2. Molecular dynamics simulations and structural modeling indicate that these mutations disrupt the lid and membrane binding domain, with Compound 2 potentially dampening these structural alterations. Conversely, variants such as M252K and F382V in the cap and α/β-hydrolase domain, respectively, exhibited limited or no rescue by Compound 2. Future research should prioritize in vivo investigations that would validate the therapeutic potential of Compound 2 and related activators in familial LCAT deficiency patients with mutations in the cap and lid of the enzyme. SIGNIFICANCE STATEMENT: Lecithin:cholesterol acyltranferase (LCAT) catalyzes the first step of reverse cholesterol transport, namely the esterification of cholesterol in high density lipoprotein particles. Somatic mutations in LCAT lead to excess cholesterol in blood plasma and, in severe cases, kidney failure. In this study, we show that recently discovered small molecule activators can rescue function in LCAT-deficient variants when the mutations occur in the lid and cap domains of the enzyme.
A novel splicing variant in ABCA1 in the first reported Hong Kong Chinese patient with high-density lipoprotein deficiency.
Low high-density lipoprotein cholesterol (HDL-C) is a risk factor for cardiovascular disease. Very low HDL-C levels (less than 20 mg/dL), however, were uncommonly seen and can be due to genetic defects involving the metabolic pathway of high-density lipoprotein (HDL). We encountered a 50-year-old Chinese man who was only noticed to have extremely low HDL-C levels after surviving recurrent episodes of myocardial infarction. Further workup revealed the undetectable level of apolipoprotein A-I, the absence of HDL on gel electrophoresis, and a novel heterozygous splicing variant in the ABCA1 gene, which was predicted to be pathogenic by in silico analysis. To the best of our knowledge, this is the first reported Hong Kong Chinese with ABCA1 deficiency and probable Tangier disease. The association of ABCA1 deficiency/Tangier disease and accelerated atherosclerosis is discussed. Clinicians should be aware of the differential diagnoses of very low HDL-C, which could be divided into genetic and acquired causes. Genetic low HDL syndromes include apoA-I deficiency, Tangier disease, and familial LCAT deficiency, each of which has characteristic clinical features and can be differentiated from the other further by apoA-I measurement, lipoprotein analysis, and genetic testing. Patients with ABCA1 deficiency and Tangier disease are at risk of premature coronary artery disease and should be aggressively screened and treated for cardiovascular risk factors and established cardiovascular diseases. Revascularization strategy and indications for coronary artery bypass grafting in patients with Tangier disease and coronary artery disease follow that as for patients without Tangier disease.
Longitudinal analysis of clinical and laboratory biomarkers in a patient with familial lecithin: cholesterol acyltransferase deficiency (FLD) and accelerated eGFR decline: A case study.
Familial lecithin:cholesterol acyltransferase (LCAT) deficiency (FLD) is an ultra-rare autosomal recessive disease characterized by very low high-density lipoprotein cholesterol (HDL-C) levels, corneal opacity, anemia, and progressive renal disease. The rate and severity of renal disease are variable across FLD patients and the biomarkers and risk factors for disease progression are poorly understood. Here we report a 30 year-long comparative analysis of the clinical and laboratory biomarkers in an FLD patient with accelerated renal decline, who underwent two kidney and one liver transplantations. Results show that elevated triglyceride and non-HDL-C levels may promote the formation of LpX and accelerate renal function decline, whereas markers of anemia may be early predictors. Conversely, corneal opacity progresses at a steady rate and does not correlate with lipid, hematologic, or renal biomarkers. Our study suggests that monitoring of markers of anemia may aid the early detection and timely management of kidney disease with conservative therapies. Furthermore, it suggests that controlling hypercholesterolemia and hypertriglyceridemia may help improve renal disease prognosis.
Publicações recentes
Lecithin-cholesterol acyltransferase deficiency with the finding of "zebra bodies": A diagnostic challenge in the context of suspected Fabry disease.
Rescue of Familial Lecithin:Cholesterol Acyltranferase Deficiency Mutations with an Allosteric Activator.
A novel splicing variant in ABCA1 in the first reported Hong Kong Chinese patient with high-density lipoprotein deficiency.
Longitudinal analysis of clinical and laboratory biomarkers in a patient with familial lecithin: cholesterol acyltransferase deficiency (FLD) and accelerated eGFR decline: A case study.
Familial LCAT Deficiency and Low HDL-C Levels: In silico Characterization of Two Rare LCAT Missense Mutations.
📚 EuropePMC36 artigos no totalmostrando 44
Lecithin-cholesterol acyltransferase deficiency with the finding of "zebra bodies": A diagnostic challenge in the context of suspected Fabry disease.
NefrologiaRescue of Familial Lecithin:Cholesterol Acyltranferase Deficiency Mutations with an Allosteric Activator.
Molecular pharmacologyA novel splicing variant in ABCA1 in the first reported Hong Kong Chinese patient with high-density lipoprotein deficiency.
Endocrinology, diabetes & metabolism case reportsLongitudinal analysis of clinical and laboratory biomarkers in a patient with familial lecithin: cholesterol acyltransferase deficiency (FLD) and accelerated eGFR decline: A case study.
Journal of clinical lipidologyFamilial LCAT Deficiency and Low HDL-C Levels: In silico Characterization of Two Rare LCAT Missense Mutations.
The application of clinical geneticsTwo novel variants in the lecithin:cholesterol acyltransferase gene resulted in classic LCAT deficiency.
Atherosclerosis plusNovel therapeutic opportunities for familial lecithin:cholesterol acyltransferase deficiency: promises and challenges.
Current opinion in lipidologyRapidly progressive renal failure to reveal LCAT deficiency in an Algerian family.
Clinical nephrologyFirst-in-human autologous implantation of genetically modified adipocytes expressing LCAT for the treatment of familial LCAT deficiency.
HeliyonA Simple Fluorescent Cholesterol Labeling Method to Cryoprotect and Detect Plasma Lipoprotein-X.
BiologyLCAT-trial-24 weeks: Protocol for a clinical study to evaluate the safety of regenerative medicine and gene therapy by the autologous transplantation of human lecithin:cholesterol acyltransferase gene-transduced human pre-adipocytes.
Contemporary clinical trials communicationsPlasma FA composition in familial LCAT deficiency indicates SOAT2-derived cholesteryl ester formation in humans.
Journal of lipid researchFamilial lecithin-cholesterol acyltransferase deficiency: If so rare, why so frequent in the state of Piauí, northeastern Brazil?
Molecular genetics and metabolism reportsLCAT- targeted therapies: Progress, failures and future.
Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapieA systematic review of the natural history and biomarkers of primary lecithin:cholesterol acyltransferase deficiency.
Journal of lipid researchThe HDL mimetic CER-001 remodels plasma lipoproteins and reduces kidney lipid deposits in inherited lecithin:cholesterol acyltransferase deficiency.
Journal of internal medicineLCAT deficiency: a systematic review with the clinical and genetic description of Mexican kindred.
Lipids in health and diseaseCurrent Status of Familial LCAT Deficiency in Japan.
Journal of atherosclerosis and thrombosisCER-001 ameliorates lipid profile and kidney disease in a mouse model of familial LCAT deficiency.
Metabolism: clinical and experimentalProgression of chronic kidney disease in familial LCAT deficiency: a follow-up of the Italian cohort.
Journal of lipid researchActivation of Naturally Occurring Lecithin:Cholesterol Acyltransferase Mutants by a Novel Activator Compound.
The Journal of pharmacology and experimental therapeuticsGenetic, biochemical, and clinical features of LCAT deficiency: update for 2020.
Current opinion in lipidologyEsterification of 4β-hydroxycholesterol and other oxysterols in human plasma occurs independently of LCAT.
Journal of lipid researchFamilial LCAT deficiency and cardiovascular disease: the game is not over. A case of dramatic multivessel atherosclerosis.
Minerva medicaLecithin:cholesterol acyltransferase: symposium on 50 years of biomedical research from its discovery to latest findings.
Journal of lipid researchNovel lecithin: cholesterol acyltransferase-based therapeutic approaches.
Current opinion in lipidologyLipid Profile Rather Than the LCAT Mutation Explains Renal Disease in Familial LCAT Deficiency.
Journal of clinical medicineIdentification and functional analysis of missense mutations in the lecithin cholesterol acyltransferase gene in a Chilean patient with hypoalphalipoproteinemia.
Lipids in health and diseaseThe P274S Mutation of Lecithin-Cholesterol Acyltransferase (LCAT) and Its Clinical Manifestations in a Large Kindred.
American journal of kidney diseases : the official journal of the National Kidney FoundationPlasma lipoprotein-X quantification on filipin-stained gels: monitoring recombinant LCAT treatment ex vivo.
Journal of lipid researchLCAT Enzyme Replacement Therapy Reduces LpX and Improves Kidney Function in a Mouse Model of Familial LCAT Deficiency.
The Journal of pharmacology and experimental therapeuticsFinding a very rare mutation in non-Caucasian LCAT patients from Southwest Asia for the first time.
Journal of cellular biochemistryMolecular basis for activation of lecithin:cholesterol acyltransferase by a compound that increases HDL cholesterol.
eLifeOcular and Genetic Characteristics Observed in Two Cases of Fish-Eye Disease.
CorneaImmune-mediated acquired lecithin-cholesterol acyltransferase deficiency: A case report and literature review.
Journal of clinical lipidologyComplete and Partial Lecithin:Cholesterol Acyltransferase Deficiency Is Differentially Associated With Atherosclerosis.
CirculationNovel Missense LCAT Gene Mutation Associated with an Atypical Phenotype of Familial LCAT Deficiency in Two Portuguese Brothers.
JIMD reportsDevelopment of a novel fluorescent activity assay for lecithin:cholesterol acyltransferase.
Annals of clinical biochemistryA case of acquired lecithin:cholesterol acyltransferase deficiency with sarcoidosis that remitted spontaneously.
CEN case reportsDepletion in LpA-I:A-II particles enhances HDL-mediated endothelial protection in familial LCAT deficiency.
Journal of lipid research[Lecithin:Cholesterol Acyltransferase Deficiency, from genes to therapy].
Giornale italiano di nefrologia : organo ufficiale della Societa italiana di nefrologiaFamilial Lecithin Cholesterol Acyl Transferase Deficiency with Chronic Kidney Disease.
The Journal of the Association of Physicians of IndiaSafety and Tolerability of ACP-501, a Recombinant Human Lecithin:Cholesterol Acyltransferase, in a Phase 1 Single-Dose Escalation Study.
Circulation researchStructure and function of lysosomal phospholipase A2 and lecithin:cholesterol acyltransferase.
Nature communicationsAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Deficiência de LCAT familiar.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Deficiência de LCAT familiar
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Lecithin-cholesterol acyltransferase deficiency with the finding of "zebra bodies": A diagnostic challenge in the context of suspected Fabry disease.
- Familial LCAT Deficiency and Low HDL-C Levels: In silico Characterization of Two Rare LCAT Missense Mutations.
- Rescue of Familial Lecithin:Cholesterol Acyltranferase Deficiency Mutations with an Allosteric Activator.
- A novel splicing variant in ABCA1 in the first reported Hong Kong Chinese patient with high-density lipoprotein deficiency.
- Longitudinal analysis of clinical and laboratory biomarkers in a patient with familial lecithin: cholesterol acyltransferase deficiency (FLD) and accelerated eGFR decline: A case study.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:79293(Orphanet)
- OMIM OMIM:245900(OMIM)
- MONDO:0009515(MONDO)
- GARD:4011(GARD (NIH))
- Variantes catalogadas(ClinVar)
- Busca completa no PubMed(PubMed)
- Q3441028(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar