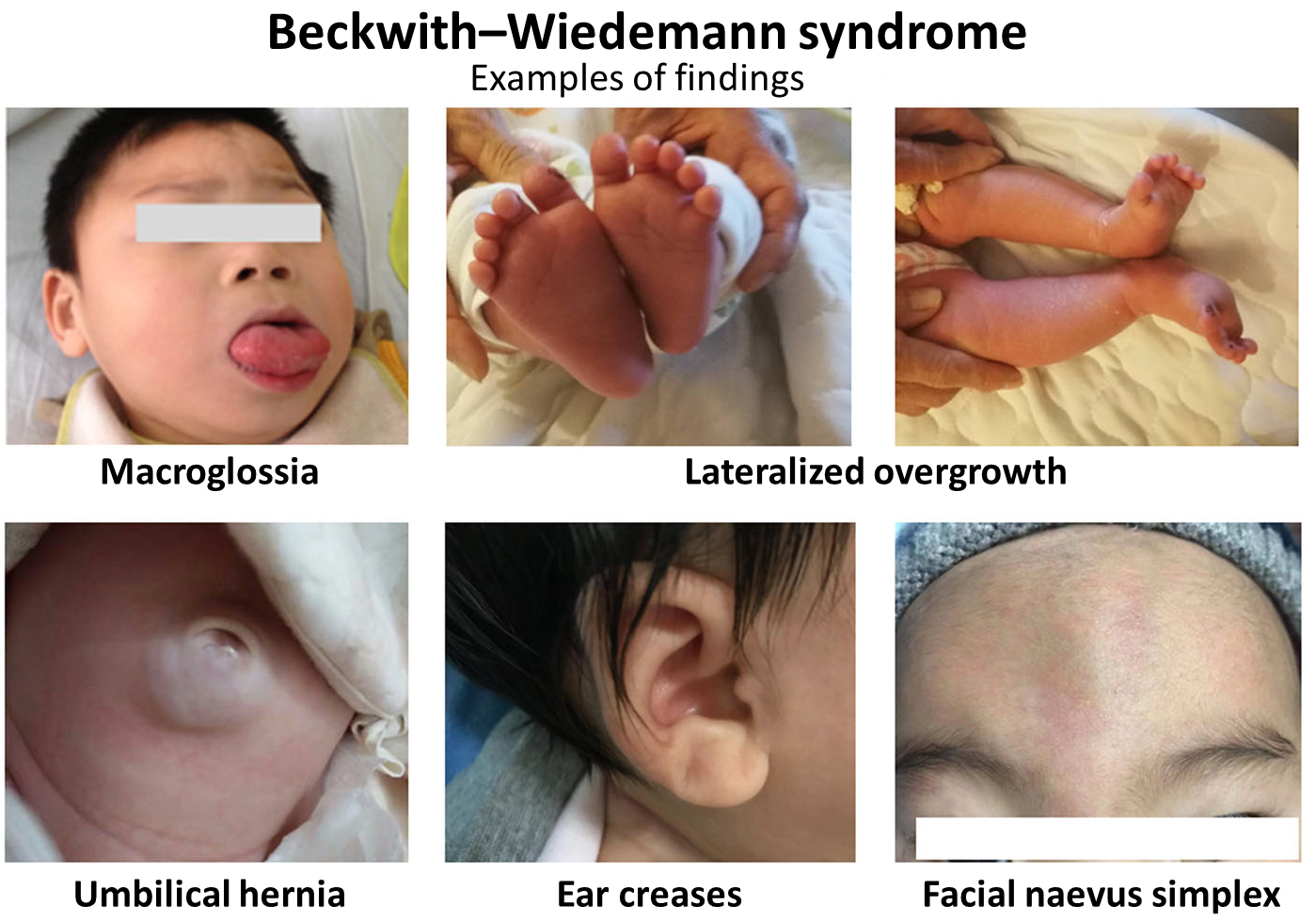
A Síndrome de Silver-Russell, causada pela dissomia uniparental materna do cromossomo 7, é uma condição genética que causa baixa estatura. Ela é caracterizada por um crescimento muito lento antes e depois do nascimento, dificuldades para se alimentar, assimetria corporal e peculiaridades no rosto e na cabeça (como rosto com formato triangular, cabeça relativamente grande, testa proeminente, queixo pequeno e cantos da boca virados para baixo). Outras características podem incluir dedo mindinho curvado, manchas na pele cor de "café com leite", alterações nos órgãos genitais masculinos, atraso leve no desenvolvimento e/ou na fala, além de problemas de movimento.
Introdução
O que você precisa saber de cara
A Síndrome de Silver-Russell, causada pela dissomia uniparental materna do cromossomo 7, é uma condição genética que causa baixa estatura. Ela é caracterizada por um crescimento muito lento antes e depois do nascimento, dificuldades para se alimentar, assimetria corporal e peculiaridades no rosto e na cabeça (como rosto com formato triangular, cabeça relativamente grande, testa proeminente, queixo pequeno e cantos da boca virados para baixo). Outras características podem incluir dedo mindinho curvado, manchas na pele cor de "café com leite", alterações nos órgãos genitais masculinos, atraso leve no desenvolvimento e/ou na fala, além de problemas de movimento.
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Partes do corpo afetadas
+ 19 sintomas em outras categorias
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 59 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Genes associados
1 gene identificado com associação a esta condição.
Adapter protein which modulates coupling of a number of cell surface receptor kinases with specific signaling pathways. Binds to, and suppress signals from, activated receptors tyrosine kinases, including the insulin (INSR) and insulin-like growth factor (IGF1R) receptors. The inhibitory effect can be achieved by 2 mechanisms: interference with the signaling pathway and increased receptor degradation. Delays and reduces AKT1 phosphorylation in response to insulin stimulation. Blocks association
Cytoplasm
Variantes genéticas (ClinVar)
11 variantes patogênicas registradas no ClinVar.
Vias biológicas (Reactome)
7 vias biológicas associadas aos genes desta condição.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Dissomia uniparental de origem materna, cromossomo 7
Centros de Referência SUS
24 centros habilitados pelo SUS para Dissomia uniparental de origem materna, cromossomo 7
Centros para Dissomia uniparental de origem materna, cromossomo 7
Detalhes dos centros
Hospital Universitário Prof. Edgard Santos (HUPES)
R. Dr. Augusto Viana, s/n - Canela, Salvador - BA, 40110-060 · CNES 0003808
Serviço de Referência
Hospital Infantil Albert Sabin
R. Tertuliano Sales, 544 - Vila União, Fortaleza - CE, 60410-794 · CNES 2407876
Serviço de Referência
Hospital de Apoio de Brasília (HAB)
AENW 3 Lote A Setor Noroeste - Plano Piloto, Brasília - DF, 70684-831 · CNES 0010456
Serviço de Referência
Hospital Estadual Infantil e Maternidade Alzir Bernardino Alves (HIABA)
Av. Min. Salgado Filho, 918 - Soteco, Vila Velha - ES, 29106-010 · CNES 6631207
Serviço de Referência
Hospital das Clínicas da UFG
Rua 235 QD. 68 Lote Área, Nº 285, s/nº - Setor Leste Universitário, Goiânia - GO, 74605-050 · CNES 2338424
Serviço de Referência
Hospital Universitário da UFJF
R. Catulo Breviglieri, Bairro - s/n - Santa Catarina, Juiz de Fora - MG, 36036-110 · CNES 2297442
Atenção Especializada
Hospital das Clínicas da UFMG
Av. Prof. Alfredo Balena, 110 - Santa Efigênia, Belo Horizonte - MG, 30130-100 · CNES 2280167
Serviço de Referência
Hospital Universitário Julio Müller (HUJM)
R. Luis Philippe Pereira Leite, s/n - Alvorada, Cuiabá - MT, 78048-902 · CNES 2726092
Atenção Especializada
Hospital Universitário João de Barros Barreto
R. dos Mundurucus, 4487 - Guamá, Belém - PA, 66073-000 · CNES 2337878
Serviço de Referência
Hospital Universitário Lauro Wanderley (HULW)
R. Tabeliao Estanislau Eloy, 585 - Castelo Branco, João Pessoa - PB, 58050-585 · CNES 0002470
Atenção Especializada
Instituto de Medicina Integral Prof. Fernando Figueira (IMIP)
R. dos Coelhos, 300 - Boa Vista, Recife - PE, 50070-902 · CNES 0000647
Serviço de Referência
Hospital Pequeno Príncipe
R. Des. Motta, 1070 - Água Verde, Curitiba - PR, 80250-060 · CNES 3143805
Serviço de Referência
Hospital Universitário Regional de Maringá (HUM)
Av. Mandacaru, 1590 - Parque das Laranjeiras, Maringá - PR, 87083-240 · CNES 2216108
Atenção Especializada
Hospital de Clínicas da UFPR
R. Gen. Carneiro, 181 - Alto da Glória, Curitiba - PR, 80060-900 · CNES 2364980
Serviço de Referência
Hospital Universitário Pedro Ernesto (HUPE-UERJ)
Blvd. 28 de Setembro, 77 - Vila Isabel, Rio de Janeiro - RJ, 20551-030 · CNES 2280221
Serviço de Referência
Instituto Nacional de Saúde da Mulher, da Criança e do Adolescente Fernandes Figueira (IFF/Fiocruz)
Av. Rui Barbosa, 716 - Flamengo, Rio de Janeiro - RJ, 22250-020 · CNES 2269988
Serviço de Referência
Hospital São Lucas da PUCRS
Av. Ipiranga, 6690 - Jardim Botânico, Porto Alegre - RS, 90610-000 · CNES 2232928
Serviço de Referência
Hospital de Clínicas de Porto Alegre (HCPA)
Rua Ramiro Barcelos, 2350 Bloco A - Av. Protásio Alves, 211 - Bloco B e C - Santa Cecília, Porto Alegre - RS, 90035-903 · CNES 2237601
Serviço de Referência
Hospital Universitário da UFSC (HU-UFSC)
R. Profa. Maria Flora Pausewang - Trindade, Florianópolis - SC, 88036-800 · CNES 2560356
Serviço de Referência
Hospital das Clínicas da FMUSP
R. Dr. Ovídio Pires de Campos, 225 - Cerqueira César, São Paulo - SP, 05403-010 · CNES 2077485
Serviço de Referência
Hospital de Base de São José do Rio Preto
Av. Brg. Faria Lima, 5544 - Vila Sao Jose, São José do Rio Preto - SP, 15090-000 · CNES 2079798
Atenção Especializada
Hospital de Clínicas da UNICAMP
R. Vital Brasil, 251 - Cidade Universitária, Campinas - SP, 13083-888 · CNES 2748223
Serviço de Referência
Hospital de Clínicas de Ribeirão Preto (HCRP-USP)
R. Ten. Catão Roxo, 3900 - Vila Monte Alegre, Ribeirão Preto - SP, 14015-010 · CNES 2082187
Serviço de Referência
UNIFESP / Hospital São Paulo
R. Napoleão de Barros, 715 - Vila Clementino, São Paulo - SP, 04024-002 · CNES 2688689
Serviço de Referência
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Ensaios em destaque
🟢 Recrutando agora
1 pesquisa recrutando participantes. Converse com seu médico sobre a possibilidade de participar.
Outros ensaios clínicos
Publicações mais relevantes
Molecular characterization of imprinting disorders: Beckwith-Wiedemann, Silver-Russell, and Prader-Willi syndromes in Egyptian patients.
Imprinted genes, characterized by monoallelic expressions (either maternal or paternal), they are crucial for normal growth and development. Disruption of their monoallelic expressions leads to imprinting disorders (ImpDis). The aim of this study is to achieve proper diagnosis of ImpDis in Egyptian patients through clinical evaluation and genetic testing, emphasizing certain clinical manifestations that may indicate ImpDis to provide accurate diagnosis and genetic counseling. Fifty-three patients, either clinically evaluated for Impaired Disposition (ImpDis) or suspected to have it, were referred from the outpatient genetic clinics at the National Research Center, Egypt. Nineteen patients displayed clinical manifestations of ImpDis syndromes, while 34 showed signs affecting growth, which suggested ImpDis. These growth-related symptoms included growth retardation, feeding problems, failure to thrive, hypoglycemia, obesity, hemihypertrophy, asymmetry, and overgrowth. Of the 19 patients with syndromic ImpDis, 8 were clinically diagnosed with Silver-Russell syndrome (SRS), 7 with Prader-Willi syndrome (PWS), and 4 with Beckwith-Wiedemann syndrome (BWS). We employed methylation-specific multiple ligation-dependent probe amplification (MS-MLPA) for all patients, SNP-array testing for 12 patients, and whole exome sequencing (WES) for one patient. In patients with Silver-Russell syndrome (SRS), one patient exhibited hypermethylation of the GRB10 and MEST genes, along with segmental uniparental disomy (UPD) on chromosome 7 (patient 1). Another patient had a variant in the HMGA2 gene (NM_001300918.1:c.310dup), which, according to the American College of Medical Genetics (ACMG) criteria, was classified as PM2 VUS (patient 2). In patients with Prader-Willi syndrome (PWS), one patient showed hypermethylation of the SNPRN gene (patient 3). In patients with Beckwith-Wiedemann syndrome (BWS), two displayed hypomethylation of the KCNQ-CR region (patients 4 and 5). Among the group of patients with symptoms suggestive of ImpDis, no methylation defects were detected through MS-MLPA. It is crucial to diagnose ImpDis accurately, as understanding the exact cause of ImpDis is important for genetic counseling and personalized medicine. Early diagnosis enables timely interventions, which can improve developmental outcomes. Precision in diagnosis helps differentiate between conditions with overlapping clinical features. HMGA2 mutation should be verified in SRs patients with negative 11p15 methylation defect and matUPD7.
Balanced Translocation t(3;12) Disrupting HMGA2 and NAALADL2 Genes in Twins With Silver-Russell Syndrome and Intellectual Disability.
Silver-Russell Syndrome (SRS) is a genetic disorder characterized by intrauterine and postnatal growth restriction. Most cases are caused by an imprinting error either with hypomethylation of the Imprinted Control Region 1 at 11p15.5, or maternal uniparental disomy of chromosome 7. Approximately 40% of the cases have unknown etiology, thus distinct mechanisms have been described in association with the syndrome. Here, we present a case of monozygotic twin sisters with a clinical diagnosis of SRS, mild intellectual disability and epilepsy who carry a balanced translocation between chromosomes 3 and 12 that interrupts the NAALADL2 and HMGA2 genes, respectively. Disruption of HMGA2, a gene previously described as causative of SRS, confirms the initial diagnosis. NAALADL2 gene has been recently proposed as a candidate for intellectual disability and could partially contribute to our patient's phenotype. Silver-Russell Syndrome (SRS) is typically characterized by gestational growth restriction resulting in affected individuals being born small for gestational age, with relative macrocephaly at birth (head circumference ≥1.5 standard deviations [SD] above birth weight and/or length), prominent forehead with frontal bossing, and frequently body asymmetry. This is typically followed by postnatal growth failure, and in some cases progressive limb length discrepancy and feeding difficulties. Additional clinical features include triangular facies, fifth finger clinodactyly, and micrognathia with narrow chin. Except for the limb length asymmetry, growth failure is proportionate and head growth typically normal. The average adult height in untreated individuals is ~3.1±1.4 SD below the mean. The Netchine-Harbison Clinical Scoring System (NH-CSS) is a sensitive diagnostic scoring system. Clinical diagnosis can be established in an individual who meets at least four of the NH-CSS clinical criteria – prominent forehead/frontal bossing and relative macrocephaly at birth plus two additional findings – and in whom other disorders have been ruled out. SRS is a genetically heterogeneous condition. Genetic testing confirms clinical diagnosis in approximately 60% of affected individuals. Hypomethylation of the imprinting control region 1 (ICR1) at 11p15.5 causes SRS in 35%-67% of individuals, and maternal uniparental disomy of chromosome 7 (upd(7)mat) causes SRS in 7%-10% of individuals. There are a small number of individuals with SRS who have duplications, deletions, or translocations involving the imprinting centers at 11p15.5 or duplications, deletions, or translocations involving chromosome 7. Rarely, affected individuals with pathogenic variants in CDKN1C, IGF2, PLAG1, and HMGA2 have been described. However, approximately 30%-40% of individuals who meet NH-CSS clinical criteria for SRS have negative molecular and/or cytogenetic testing. Treatment of manifestations: Multidisciplinary follow up and early specific intervention are necessary for optimal management of affected individuals, including early referral to an endocrinologist. Treatment may include growth hormone therapy. Hypoglycemia should be prevented or aggressively managed. Strategies for feeding issues include nutritional and caloric supplementation, medication for gastroesophageal reflux, therapy for oral motor problems and oral aversion, cyproheptadine for appetite stimulation, and enteral tube feeding as needed. Lower limb length discrepancy exceeding 2 cm requires intervention. In the majority of affected older children, distraction osteogenesis is recommended. Severe micrognathia or cleft palate should be managed by a multidisciplinary craniofacial team. Males with cryptorchidism or hypospadias should be referred to a urologist. Males with micropenis and females with internal genitourinary anomalies benefit from a referral to a multidisciplinary disorders of sex development (DSD) center. Physical, occupational, speech, and language therapy with an individualized education plan are used to treat developmental delays. Psychological counseling can be used as needed to address psychosocial and body image issues. Surveillance: Monitoring of growth velocity, blood glucose concentration, and urine ketones for hypoglycemia in infants and as needed in older children; evaluation of nutritional status and oral intake at each visit as well as managing tube feedings if needed; limb length assessment at each well child visit in early childhood for evidence of asymmetric growth; evaluation for scoliosis, signs of precocious puberty, genitourinary issues, dental crowding and malocclusion, and speech-language development at each visit. Agents/circumstances to avoid: Prolonged fasting in infants and young children because of the risk for hypoglycemia; elective surgery whenever possible due to risk of hypoglycemia, hypothermia, difficult healing, and difficult intubation. Risk to family members: In most families, a proband with SRS represents a simplex case (a single affected family member) and has SRS as a result of an apparent de novo epigenetic or genetic alteration. While the majority of families are presumed to have a very low recurrence risk, SRS can occur as the result of a genetic alteration associated with up to a 50% recurrence risk depending on the nature of the genetic alteration and the sex of the transmitting parent. Rare familial cases of SRS have been reported with several underlying mechanisms, including maternally inherited 11p15 duplications, maternally inherited CDKN1C gain-of-function pathogenic variants, paternally inherited IGF2 loss-of-function pathogenic variants, paternally inherited small deletions close to the boundaries of the ICR1, and paternally or maternally inherited deletions and intragenic pathogenic variants involving PLAG1 or HMGA2. Reliable SRS recurrence risk assessment therefore requires identification of the causative genetic mechanism in the proband. Prenatal testing: Reliable prenatal testing for loss of paternal methylation at the 11p1.5 ICR1 H19/IGF2 region is not possible. Prenatal testing for the following SRS-related genetic mechanisms is possible provided the genetic mechanism has been demonstrated to be causative in an affected family member: upd(7)mat, SRS-related chromosomal abnormalities, intragenic CDKN1C, IGF2, HMGA2, or PLAG1 pathogenic variants, and deletions involving HMGA2 or PLAG1. The prenatal finding of a genetic alteration consistent with SRS cannot be used to reliably predict clinical outcome because children with SRS demonstrate varying responses to growth hormone, variable late catch-up growth, and variable developmental outcomes.
Rare Causes and Differential Diagnosis in Patients With Silver-Russell Syndrome.
Silver-Russell syndrome (SRS) is an imprinting disorder mainly characterized by pre- and postnatal growth restriction. Most SRS cases are due to 11p15.5 loss of methylation (11p15.5 LOM) or maternal uniparental disomy of chromosome 7 [UPD(7)mat], but several patients remain molecularly undiagnosed. This study describes the molecular investigation of children with a clinical diagnosis or suspicion of SRS at a tertiary center specialized in growth disorders. Thirty-nine patients were evaluated with multiplex ligation-dependent probe amplification, chromosomal microarray and/or massively parallel sequencing. The most common result was 11p15.5 LOM (n = 17; 43.5%), followed by UPD(7)mat (n = 2; 5.1%). Additionally, we found maternal duplications of the imprinting centers in 11p15.5 (n = 2; 5.1%), and genetic defects in SRS-causing genes (IGF2 and HMGA2) (n = 3; 7.7%; two mutations and one deletion). Alternative molecular diagnoses included UPD(14)mat (n = 1; 2,6%), UPD(20)mat (n = 1;2,6%), copy number variants (n = 2; 5.1%), and mutations in genes associated with other growth disorders (n = 4; 10.3%), leading to diagnoses of Temple syndrome, Mulchandani-Bhoj-Conlin syndrome, IGF-1 resistance (IGF1R), Bloom syndrome (BLM), Gabriele-De Vries syndrome (YY1), Intellectual developmental disorder autosomal dominant 50 with behavioral abnormalities (NAA15), and Intellectual developmental disorder 64 (ZNF292). These findings underscore the importance of establishing the molecular diagnosis of SRS and its differential diagnoses to guide appropriate management and genetic counseling.
PEG10 loss of function causes Silver-Russell syndrome: a familial case with paternal deletion.
Silver-Russell syndrome (SRS, MIM#180860) is an imprinting disorder characterized by prenatal and postnatal growth retardation, relative macrocephaly at birth, prominent forehead, feeding difficulties, and body asymmetry. Clinical diagnosis is based on at least 4 out of 6 clinical signs (Netchine-Harbison clinical scoring system). The main molecular mechanisms are loss of methylation at the paternal H19/IGF2:IG-DMR (30-60%) at the 11p15 chromosomal region and maternal uniparental disomy of chromosome 7 (5-10%). While it is well known that deregulation of 11p15 imprinted genes (IGF2, H19, and CDKN1C) contributes to the SRS phenotype, those on chromosome 7 (GRB10, PEG10, and MEST) are still in discussion. We report two brothers clinically diagnosed with SRS (postnatal growth delay, relative macrocephaly at birth, feeding difficulties, and SRS facies). One patient also has distal tremors and a treated growth hormone deficiency. CGH-array revealed a deletion of 109 Kb including PEG10 and SGCE genes, inherited from their unaffected father. Whole Exome Sequencing did not disclose other causative variants. PEG10 functions as a transcriptional repressor of cyclin-dependent kinase inhibitors, including CDKN1C, for which maternal gain-of-function variants are linked to SRS. Real-time PCR studies showed a downregulation of PEG10 and an upregulation of CDKN1C expression only in the affected brothers. Interestingly, IGF2 was upregulated in the patient 1 under GH administration. Our findings provide the first evidence supporting the role of PEG10 in the pathogenesis of Silver-Russell Syndrome, mediated by a gain-of-function effect on the CDKN1C expression, offering new insights into the molecular mechanisms underlying this condition. The online version contains supplementary material available at 10.1038/s41598-025-32983-y.
Hypomethylation of the MEG8:Int2-DMR in patients with pathogenic PLAG1 variants suggests new role of the chr14q32 imprinting cluster in Silver-Russell syndrome.
Silver-Russell syndrome (SRS) is a clinically and genetically heterogeneous imprinting disorder. The most common molecular defects are loss of methylation of the H19/IGF2:IG-DMR on chromosome 11p15.5, followed by maternal uniparental disomy of chromosome 7. Further molecular lesions are genetic variants in the PLAG1 oncogene, as well as in the transcription factor HMGA2 and the fetal growth factor IGF2. A phenotypic overlap exists between SRS and Temple syndrome (TS14) that is also characterized by growth restriction but associated with abnormalities in the imprinted chromosome 14q32 gene cluster. In TS14 patients, the germline MEG3/DLK1: IG-DMR is hypomethylated and the MEG8:Int2-DMR gains methylation probably as consequence of transcriptional readthrough from the MEG3 promoter on the paternal chromosome. However, the functional role of the MEG8 DMR remains unknown. We analysed the DNA methylation of 11-12 imprinted regions in 17 cases with clinical SRS features and heterozygous for a PLAG1 variant. We observed a specific loss of methylation of the MEG8:Int2-DMR in the ten cases carrying pathogenic PLAG1 variants that result in stable aberrant proteins. Normal MEG8 methylation was observed in the cases carrying variants of uncertain pathogenicity or gene deletions. Most of the PLAG1 cases are familial and both epigenetic and genetic defects co-segregated within the families. Additionally, we assessed the methylation status of the MEG8:Int2-DMR in several SRS patients with HMGA2 or IGF2 variants, H19/IGF2:IG-DMR-LoM and upd(7)mat and all of them showed normal methylation. Our results indicate that pathogenic PLAG1 variants leading to stable aberrant PLAG1 proteins and possibly acting in a dominant-negative manner influence methylation of the MEG8 locus. This study suggests a new pathogenetic mechanism of the PLAG1 gene in SRS, involving imprinted genes in the chr14q32 cluster through deregulation of the MEG8:Int2-DMR and provides an epigenetic signature that may be used to assess the damaging potential of the PLAG1 variants.
Publicações recentes
Investigation of methylation profiles in Silver-Russell syndrome to explore episignatures.
Maternal uniparental disomy of chromosome 7: how chromosome 7-encoded imprinted genes contribute to the Silver-Russell phenotype.
Rare Causes and Differential Diagnosis in Patients With Silver-Russell Syndrome.
A 79-kb paternally inherited 7q32.2 microdeletion involving MEST in a patient with a Silver-Russell syndrome-like phenotype.
Segmental maternal uniparental disomy of chromosome 7q in a patient with congenital chloride diarrhea.
📚 EuropePMCmostrando 64
PEG10 loss of function causes Silver-Russell syndrome: a familial case with paternal deletion.
Scientific reportsHypomethylation of the MEG8:Int2-DMR in patients with pathogenic PLAG1 variants suggests new role of the chr14q32 imprinting cluster in Silver-Russell syndrome.
Clinical epigeneticsInvestigation of methylation profiles in Silver-Russell syndrome to explore episignatures.
Clinical epigeneticsMolecular characterization of imprinting disorders: Beckwith-Wiedemann, Silver-Russell, and Prader-Willi syndromes in Egyptian patients.
BMC pediatricsFunctional Independence of Taiwanese Children with Silver-Russell Syndrome.
Diagnostics (Basel, Switzerland)Maternal uniparental disomy of chromosome 7: how chromosome 7-encoded imprinted genes contribute to the Silver-Russell phenotype.
Clinical epigeneticsPrenatal Detection of Silver-Russell Syndrome: A First Trimester Suspicion and Diagnostic Approach.
Medicina (Kaunas, Lithuania)Balanced Translocation t(3;12) Disrupting HMGA2 and NAALADL2 Genes in Twins With Silver-Russell Syndrome and Intellectual Disability.
Clinical geneticsRare Causes and Differential Diagnosis in Patients With Silver-Russell Syndrome.
Clinical geneticsApproach to the Patient With Suspected Silver-Russell Syndrome.
The Journal of clinical endocrinology and metabolism11p13 microduplication: a differential diagnosis of Silver-Russell syndrome?
Molecular cytogeneticsDeep Brain Stimulation for an Unusual Presentation of Myoclonus Dystonia Associated with Russell-Silver Syndrome.
Tremor and other hyperkinetic movements (New York, N.Y.)Incidental finding at methylation-specific multiplex ligation-dependent probe amplification (MS-MLPA): how to proceed?
Frontiers in geneticsClinical characterization of PLAG1- related Silver-Russell syndrome:A clinical report.
European journal of medical geneticsThe effects of 3-year growth hormone treatment and body composition in Polish patients with Silver-Russell syndrome.
Endokrynologia PolskaExecutive functioning in adolescents and adults with Silver-Russell syndrome.
PloS oneMaternal uniparental disomy of chromosome 7 underlying argininosuccinic aciduria and Silver-Russell syndrome.
Human genome variationPubertal timing in children with Silver Russell syndrome compared to those born small for gestational age.
Frontiers in endocrinologyFrequency and clinical characteristics of distinct etiologies in patients with Silver-Russell syndrome diagnosed based on the Netchine-Harbison clinical scoring system.
Journal of human geneticsA 79-kb paternally inherited 7q32.2 microdeletion involving MEST in a patient with a Silver-Russell syndrome-like phenotype.
American journal of medical genetics. Part AMicrodeletions in 1q21 and 8q12.1 depict two additional molecular subgroups of Silver-Russell syndrome like phenotypes.
Molecular cytogeneticsClinical and Molecular Heterogeneity of Silver-Russell Syndrome and Therapeutic Challenges: A Systematic Review.
Current pediatric reviewsEpigenotype, Genotype, and Phenotype Analysis of Taiwanese Patients with Silver-Russell Syndrome.
Journal of personalized medicineAcquired uniparental disomy of chromosome 7 in a patient with MIRAGE syndrome that veiled a pathogenic SAMD9 variant.
Clinical pediatric endocrinology : case reports and clinical investigations : official journal of the Japanese Society for Pediatric EndocrinologyScreening of patients born small for gestational age with the Silver-Russell syndrome phenotype for DLK1 variants.
European journal of human genetics : EJHGFurther heterogeneity in Silver-Russell syndrome: PLAG1 deletion in association with a complex chromosomal rearrangement.
American journal of medical genetics. Part ASegmental maternal uniparental disomy of chromosome 7q in a patient with congenital chloride diarrhea.
Journal of clinical laboratory analysisMaternal Uniparental Disomy of Chromosome 20 (UPD(20)mat) as Differential Diagnosis of Silver Russell Syndrome: Identification of Three New Cases.
GenesChronic Hypercapnic Respiratory Failure in an Adult Patient with Silver-Russell Syndrome.
Internal medicine (Tokyo, Japan)One test for all: whole exome sequencing significantly improves the diagnostic yield in growth retarded patients referred for molecular testing for Silver-Russell syndrome.
Orphanet journal of rare diseasesNovel Variant in PLAG1 in a Familial Case with Silver-Russell Syndrome Suspicion.
GenesRecombinant Chromosome 7 Driven by Maternal Chromosome 7 Pericentric Inversion in a Girl with Features of Silver-Russell Syndrome.
International journal of molecular sciencesMaternal UPD of chromosome 7 in a patient with Silver-Russell syndrome and Pendred syndrome.
Journal of clinical laboratory analysisContribution of gene mutations to Silver-Russell syndrome phenotype: multigene sequencing analysis in 92 etiology-unknown patients.
Clinical epigeneticsPhenotype of genetically confirmed Silver-Russell syndrome beyond childhood.
Journal of medical geneticsRare De Novo IGF2 Variant on the Paternal Allele in a Patient With Silver-Russell Syndrome.
Frontiers in geneticsCognitive Profiles and Brain Volume Are Affected in Patients with Silver-Russell Syndrome.
The Journal of clinical endocrinology and metabolismSleep disordered breathing in Silver-Russell syndrome patients: a new outcome.
Sleep medicineAutistic traits and cognitive abilities associated with two molecular causes of Silver-Russell syndrome.
Journal of abnormal psychologyContribution of GRB10 to the prenatal phenotype in Silver-Russell syndrome? Lessons from 7p12 copy number variations.
European journal of medical geneticsCri-du-chat syndrome mimics Silver-Russell syndrome depending on the size of the deletion: a case report.
BMC medical genomicsSegmental Maternal UPD of Chromosome 7q in a Patient With Pendred and Silver Russell Syndromes-Like Features.
Frontiers in geneticsA case report and review of the literature indicate that HMGA2 should be added as a disease gene for Silver-Russell syndrome.
GeneClinical Manifestations and Metabolic Outcomes of Seven Adults With Silver-Russell Syndrome.
The Journal of clinical endocrinology and metabolismThe effects of growth hormone therapy on the somatic development of a group of Polish children with Silver-Russell syndrome.
Neuro endocrinology lettersStructural and sequence variants in patients with Silver-Russell syndrome or similar features-Curation of a disease database.
Human mutationHypomethylation of HOXA4 promoter is common in Silver-Russell syndrome and growth restriction and associates with stature in healthy children.
Scientific reportsMosaic UPD(7q)mat in a patient with silver Russell syndrome.
Molecular cytogeneticsCystic fibrosis and Silver-Russell syndrome due to a partial maternal isodisomy of chromosome 7.
Clinical case reportsFormation of upd(7)mat by trisomic rescue: SNP array typing provides new insights in chromosomal nondisjunction.
Molecular cytogeneticsThe Diagnostic Value of IGF-2 and the IGF/IGFBP-3 System in Silver-Russell Syndrome.
Hormone research in paediatricsGenome-wide analysis of differential DNA methylation in Silver-Russell syndrome.
Science China. Life sciencesGenetic heterogeneity of patients with suspected Silver-Russell syndrome: genome-wide copy number analysis in 82 patients without imprinting defects.
Clinical epigeneticsSilver-Russell syndrome in Hong Kong.
Hong Kong medical journal = Xianggang yi xue za zhiNew clinical and molecular insights into Silver-Russell syndrome.
Current opinion in pediatricsNSD1 duplication in Silver-Russell syndrome (SRS): molecular karyotyping in patients with SRS features.
Clinical geneticsNew developments in Silver-Russell syndrome and implications for clinical practice.
EpigenomicsLong-Term Results of GH Treatment in Silver-Russell Syndrome (SRS): Do They Benefit the Same as Non-SRS Short-SGA?
The Journal of clinical endocrinology and metabolismMicrodeletions of the 7q32.2 imprinted region are associated with Silver-Russell syndrome features.
American journal of medical genetics. Part ASilver-Russell Syndrome - Part I: Clinical Characteristics and Genetic Background.
Pediatric endocrinology, diabetes, and metabolismPrenatal molecular testing for Beckwith-Wiedemann and Silver-Russell syndromes: a challenge for molecular analysis and genetic counseling.
European journal of human genetics : EJHGA splicing mutation of the HMGA2 gene is associated with Silver-Russell syndrome phenotype.
Journal of human geneticsPrevalence and management of gastrointestinal manifestations in Silver-Russell syndrome.
Archives of disease in childhoodGenome-wide methylation analysis in Silver-Russell syndrome patients.
Human geneticsAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Dissomia uniparental de origem materna, cromossomo 7.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Dissomia uniparental de origem materna, cromossomo 7
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Molecular characterization of imprinting disorders: Beckwith-Wiedemann, Silver-Russell, and Prader-Willi syndromes in Egyptian patients.
- Balanced Translocation t(3;12) Disrupting HMGA2 and NAALADL2 Genes in Twins With Silver-Russell Syndrome and Intellectual Disability.
- Rare Causes and Differential Diagnosis in Patients With Silver-Russell Syndrome.
- PEG10 loss of function causes Silver-Russell syndrome: a familial case with paternal deletion.
- Hypomethylation of the MEG8:Int2-DMR in patients with pathogenic PLAG1 variants suggests new role of the chr14q32 imprinting cluster in Silver-Russell syndrome.
- Investigation of methylation profiles in Silver-Russell syndrome to explore episignatures.
- Maternal uniparental disomy of chromosome 7: how chromosome 7-encoded imprinted genes contribute to the Silver-Russell phenotype.
- A 79-kb paternally inherited 7q32.2 microdeletion involving MEST in a patient with a Silver-Russell syndrome-like phenotype.
- Segmental maternal uniparental disomy of chromosome 7q in a patient with congenital chloride diarrhea.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:96182(Orphanet)
- MONDO:0019913(MONDO)
- GARD:19334(GARD (NIH))
- Variantes catalogadas(ClinVar)
- Busca completa no PubMed(PubMed)
- Q55788987(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar