Introdução
O que você precisa saber de cara
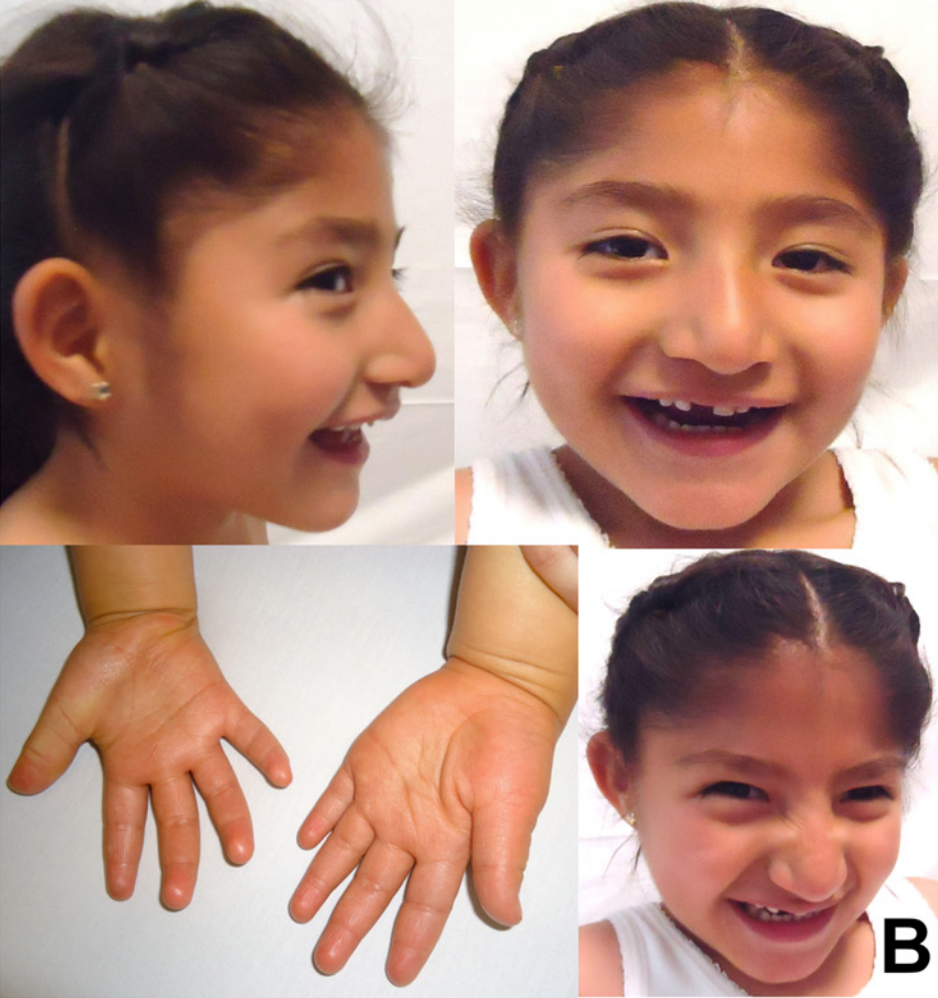
A síndrome de Angelman (SA) é um distúrbio genético raro que afeta aproximadamente 1 em cada 15.000 indivíduos. A SA prejudica o funcionamento do sistema nervoso, produzindo sintomas como deficiência intelectual grave, deficiência no desenvolvimento, fala funcional limitada ou ausente, problemas de equilíbrio e movimento, convulsões, hiperatividade e problemas de sono. Os sintomas físicos incluem cabeça pequena e uma aparência facial específica. Além disso, os indivíduos afetados costumam ter uma personalidade alegre. A síndrome de Angelman envolve genes que também foram associados a 1–2% dos casos de transtorno do espectro autista.
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Entender a doença
Do básico ao detalhe, leia no seu ritmo
Preparando trilha educativa...
Sinais e sintomas
O que aparece no corpo e com que frequência cada sintoma acontece
Partes do corpo afetadas
+ 14 sintomas em outras categorias
Características mais comuns
Os sintomas variam de pessoa para pessoa. Abaixo estão as 27 características clínicas mais associadas, ordenadas por frequência.
Linha do tempo da pesquisa
Encontrou um erro ou informação desatualizada? Sugira uma correção →
Genética e causas
O que está alterado no DNA e como passa nas famílias
Genes associados
3 genes identificados com associação a esta condição.
Catalytic component of P4-ATPase flippase complex, which catalyzes the hydrolysis of ATP coupled to the transport of phosphatidylcholine (PC) from the outer to the inner leaflet of the plasma membrane (PubMed:25947375, PubMed:29599178, PubMed:30530492). Initiates inward plasma membrane bending and recruitment of Bin/amphiphysin/Rvs (BAR) domain-containing proteins involved in membrane tubulation and cell trafficking (PubMed:29599178). Facilitates ITGB1/beta1 integrin endocytosis, delaying cell a
Cell membraneEndoplasmic reticulum membrane
May be involved in tissue-specific alternative RNA processing events
Nucleus
E3 ubiquitin-protein ligase which accepts ubiquitin from an E2 ubiquitin-conjugating enzyme in the form of a thioester and transfers it to its substrates (PubMed:10373495, PubMed:16772533, PubMed:19204938, PubMed:19233847, PubMed:19325566, PubMed:19591933, PubMed:22645313, PubMed:24273172, PubMed:24728990, PubMed:30020076). Several substrates have been identified including the BMAL1, ARC, LAMTOR1, RAD23A and RAD23B, MCM7 (which is involved in DNA replication), annexin A1, the PML tumor suppresso
CytoplasmNucleus
Angelman syndrome
A neurodevelopmental disorder characterized by severe motor and intellectual retardation, ataxia, frequent jerky limb movements and flapping of the arms and hands, hypotonia, seizures, absence of speech, frequent smiling and episodes of paroxysmal laughter, open-mouthed expression revealing the tongue.
Variantes genéticas (ClinVar)
1,412 variantes patogênicas registradas no ClinVar.
Vias biológicas (Reactome)
5 vias biológicas associadas aos genes desta condição.
Diagnóstico
Os sinais que médicos procuram e os exames que confirmam
Tratamento e manejo
Remédios, cuidados de apoio e o que precisa acompanhar
Onde tratar no SUS
Hospitais de referência no Brasil e o protocolo oficial do SUS (PCDT)
🇧🇷 Atendimento SUS — Síndrome Angelman por anomalia do imprinting no locus 15q11-q13
Centros de Referência SUS
24 centros habilitados pelo SUS para Síndrome Angelman por anomalia do imprinting no locus 15q11-q13
Centros para Síndrome Angelman por anomalia do imprinting no locus 15q11-q13
Detalhes dos centros
Hospital Universitário Prof. Edgard Santos (HUPES)
R. Dr. Augusto Viana, s/n - Canela, Salvador - BA, 40110-060 · CNES 0003808
Serviço de Referência
Hospital Infantil Albert Sabin
R. Tertuliano Sales, 544 - Vila União, Fortaleza - CE, 60410-794 · CNES 2407876
Serviço de Referência
Hospital de Apoio de Brasília (HAB)
AENW 3 Lote A Setor Noroeste - Plano Piloto, Brasília - DF, 70684-831 · CNES 0010456
Serviço de Referência
Hospital Estadual Infantil e Maternidade Alzir Bernardino Alves (HIABA)
Av. Min. Salgado Filho, 918 - Soteco, Vila Velha - ES, 29106-010 · CNES 6631207
Serviço de Referência
Hospital das Clínicas da UFG
Rua 235 QD. 68 Lote Área, Nº 285, s/nº - Setor Leste Universitário, Goiânia - GO, 74605-050 · CNES 2338424
Serviço de Referência
Hospital Universitário da UFJF
R. Catulo Breviglieri, Bairro - s/n - Santa Catarina, Juiz de Fora - MG, 36036-110 · CNES 2297442
Atenção Especializada
Hospital das Clínicas da UFMG
Av. Prof. Alfredo Balena, 110 - Santa Efigênia, Belo Horizonte - MG, 30130-100 · CNES 2280167
Serviço de Referência
Hospital Universitário Julio Müller (HUJM)
R. Luis Philippe Pereira Leite, s/n - Alvorada, Cuiabá - MT, 78048-902 · CNES 2726092
Atenção Especializada
Hospital Universitário João de Barros Barreto
R. dos Mundurucus, 4487 - Guamá, Belém - PA, 66073-000 · CNES 2337878
Serviço de Referência
Hospital Universitário Lauro Wanderley (HULW)
R. Tabeliao Estanislau Eloy, 585 - Castelo Branco, João Pessoa - PB, 58050-585 · CNES 0002470
Atenção Especializada
Instituto de Medicina Integral Prof. Fernando Figueira (IMIP)
R. dos Coelhos, 300 - Boa Vista, Recife - PE, 50070-902 · CNES 0000647
Serviço de Referência
Hospital Pequeno Príncipe
R. Des. Motta, 1070 - Água Verde, Curitiba - PR, 80250-060 · CNES 3143805
Serviço de Referência
Hospital Universitário Regional de Maringá (HUM)
Av. Mandacaru, 1590 - Parque das Laranjeiras, Maringá - PR, 87083-240 · CNES 2216108
Atenção Especializada
Hospital de Clínicas da UFPR
R. Gen. Carneiro, 181 - Alto da Glória, Curitiba - PR, 80060-900 · CNES 2364980
Serviço de Referência
Hospital Universitário Pedro Ernesto (HUPE-UERJ)
Blvd. 28 de Setembro, 77 - Vila Isabel, Rio de Janeiro - RJ, 20551-030 · CNES 2280221
Serviço de Referência
Instituto Nacional de Saúde da Mulher, da Criança e do Adolescente Fernandes Figueira (IFF/Fiocruz)
Av. Rui Barbosa, 716 - Flamengo, Rio de Janeiro - RJ, 22250-020 · CNES 2269988
Serviço de Referência
Hospital São Lucas da PUCRS
Av. Ipiranga, 6690 - Jardim Botânico, Porto Alegre - RS, 90610-000 · CNES 2232928
Serviço de Referência
Hospital de Clínicas de Porto Alegre (HCPA)
Rua Ramiro Barcelos, 2350 Bloco A - Av. Protásio Alves, 211 - Bloco B e C - Santa Cecília, Porto Alegre - RS, 90035-903 · CNES 2237601
Serviço de Referência
Hospital Universitário da UFSC (HU-UFSC)
R. Profa. Maria Flora Pausewang - Trindade, Florianópolis - SC, 88036-800 · CNES 2560356
Serviço de Referência
Hospital das Clínicas da FMUSP
R. Dr. Ovídio Pires de Campos, 225 - Cerqueira César, São Paulo - SP, 05403-010 · CNES 2077485
Serviço de Referência
Hospital de Base de São José do Rio Preto
Av. Brg. Faria Lima, 5544 - Vila Sao Jose, São José do Rio Preto - SP, 15090-000 · CNES 2079798
Atenção Especializada
Hospital de Clínicas da UNICAMP
R. Vital Brasil, 251 - Cidade Universitária, Campinas - SP, 13083-888 · CNES 2748223
Serviço de Referência
Hospital de Clínicas de Ribeirão Preto (HCRP-USP)
R. Ten. Catão Roxo, 3900 - Vila Monte Alegre, Ribeirão Preto - SP, 14015-010 · CNES 2082187
Serviço de Referência
UNIFESP / Hospital São Paulo
R. Napoleão de Barros, 715 - Vila Clementino, São Paulo - SP, 04024-002 · CNES 2688689
Serviço de Referência
Dados de DATASUS/CNES, SBGM, ABNeuro e Ministério da Saúde. Sempre confirme a disponibilidade diretamente com o estabelecimento.
Pesquisa ativa
Ensaios clínicos abertos e novidades científicas recentes
Pesquisa e ensaios clínicos
Nenhum ensaio clínico registrado para esta condição.
Publicações mais relevantes
Clinical Presentation, Genetics, and Laboratory Testing with Integrated Genetic Analysis of Molecular Mechanisms in Prader-Willi and Angelman Syndromes: A Review.
Prader-Willi (PWS) and Angelman (AS) syndromes were the first examples in humans with errors in genomic imprinting, usually from de novo 15q11-q13 deletions of different parent origin (paternal in PWS and maternal in AS). Dozens of genes and transcripts are found in the 15q11-q13 region, and may play a role in PWS, specifically paternally expressed SNURF-SNRPN and MAGEL2 genes, while AS is due to the maternally expressed UBE3A gene. These three causative genes, including their encoding proteins, were targeted. This review article summarizes and illustrates the current understanding and cause of both PWS and AS using strategies to include the literature sources of key words and searchable web-based programs with databases for integrated gene and protein interactions, biological processes, and molecular mechanisms available for the two imprinting disorders. The SNURF-SNRPN gene is key in developing complex spliceosomal snRNP assemblies required for mRNA processing, cellular events, splicing, and binding required for detailed protein production and variation, neurodevelopment, immunodeficiency, and cell migration. The MAGEL2 gene is involved with the regulation of retrograde transport and promotion of endosomal assembly, oxytocin and reproduction, as well as circadian rhythm, transcriptional activity control, and appetite. The UBE3A gene encodes a key enzyme for the ubiquitin protein degradation system, apoptosis, tumor suppression, cell adhesion, and targeting proteins for degradation, autophagy, signaling pathways, and circadian rhythm. PWS is characterized early with infantile hypotonia, a poor suck, and failure to thrive with hypogenitalism/hypogonadism. Later, growth and other hormone deficiencies, developmental delays, and behavioral problems are noted with hyperphagia and morbid obesity, if not externally controlled. AS is characterized by seizures, lack of speech, severe learning disabilities, inappropriate laughter, and ataxia. This review captures the clinical presentation, natural history, causes with genetics, mechanisms, and description of established laboratory testing for genetic confirmation of each disorder. Three separate searchable web-based programs and databases that included information from the updated literature and other sources were used to identify and examine integrated genetic findings with predicted gene and protein interactions, molecular mechanisms and functions, biological processes, pathways, and gene-disease associations for candidate or causative genes per disorder. The natural history, review of pathophysiology, clinical presentation, genetics, and genetic-phenotypic findings were described along with computational biology, molecular mechanisms, genetic testing approaches, and status for each disorder, management and treatment options, clinical trial experiences, and future strategies. Conclusions and limitations were discussed to improve understanding, clinical care, genetics, diagnostic protocols, therapeutic agents, and genetic counseling for those with these genomic imprinting disorders.
UBE3A reinstatement restores behaviorand proteome in an Angelman syndrome mouse model of imprinting defects.
Angelman Syndrome (AS) is a severe neurodevelopmental disorder with only symptomatic treatment currently available. The primary cause of AS is loss of functional UBE3A protein. This can be caused by deletions in the maternal 15q11-q13 region, maternal AS-imprinting center defects (mICD), paternal uniparental disomy of chromosome 15 (UPD) or mutations within the UBE3A gene. Current mouse models are Ube3a-centric and do not address expression changes of other genes in the 15q11-q13 locus on the pathophysiology of AS. This limits the ability to discern differences in therapeutic responses to current UBE3A-targeting strategies and hampers the identification of novel therapeutics/co-therapeutics. Using a mouse line that harbors a maternally inherited mutation affecting the AS-PWS imprinting center ('mICD mice'), we studied the impact of the mICD or UPD AS subtype on behavior, seizure susceptibility and proteome. Additionally, by using mice overexpressing two copies of Ube3a or antisense oligonucleotide (ASO) targeting Ube3a-ATS, we analyzed the impact of bi-allelic Ube3a activation on behavior and proteome. mICD mice showed 80% reduction in UBE3A protein, bi-allelic expression of Ube3a-ATS and Mkrn3-Snord115 gene cluster, leading to robust AS behavioral deficits and proteome alterations similar to Ube3am-/p+ mice. Genetic UBE3A overexpression in mICD mice, mimicking therapeutic strategies that effectively activate the biallelic silenced Ube3a gene, resulted in a complete rescue of all behavioral phenotypes, seizure susceptibility and proteome alterations. Subsequently, treatment with an antisense oligonucleotide (ASO) to directly activate the biallelic silenced Ube3a gene in mICD mice also resulted in efficient reinstatement of UBE3A, 30% higher relative to WT, alongside a partial rescue of behavioral phenotypes. Despite using a highly robust AS-specific behavioral battery, we did not investigate readouts such as neuronal activity and sleep, for which impairments in Ube3am-/p+ mice were described. Taken together, these findings demonstrate that the loss of UBE3A protein is the primary factor underlying AS phenotypes in this mICD/UPD mouse model of AS, while the biallelic expressed genes in this locus play either a marginal or yet unidentified role. These findings also corroborate UBE3A reinstatement as an attractive therapeutic strategy for AS individuals carrying an mICD or UPD mutation.
Relationship of thyroid function with genetic subtypes and treatment with growth hormone in Prader-Willi syndrome.
Prader-Willi syndrome (PWS) is the most common genetic syndrome with obesity and results from loss of expression of paternally inherited genes on chromosome 15q11-q13 by a variety of mechanisms which include large deletions (70%-75%), maternal uniparental disomy (UPD) (20%-30%), and imprinting defects (2%-5%) or balanced translocations. Individuals often have a characteristic behavior disorder with mild intellectual disability, infantile hypotonia associated with poor sucking, short stature, and obesity. PWS is characterized by hypothalamic-pituitary axis dysfunction with growth hormone (GH) deficiency, hypogonadism, and several other hormonal deficiencies resulting in short stature, centrally driven excessive appetite (hyperphagia), central obesity, cryptorchidism, and decreased lean body mass. In this study, we determined and sought differences in the incidence of thyroid abnormalities among the common genetic subtypes in a cohort of 52 subjects with PWS because there was limited literature available. We also sought the effects of growth hormone (GH) treatment on the thyroid profile. Fifty-two subjects with a genetically confirmed diagnosis of PWS were included in this study at the University of California, Irvine. Blood samples for baseline thyroxine stimulating hormone (TSH) and free thyroxine (fT4) levels were obtained in the morning after an overnight fast for 8-12 h. Statistical analyses were performed with SPSS (SPSS Inc., 21.0). Mean values were analyzed by one-way ANOVA, and student's t-test and statistical significance were set at p < 0.05. The subjects included 26 males and 26 females with an age range of 3-38 years. There were 29 subjects with chromosome 15q11-q13 deletions and 23 with UPD; 28 were GH treated currently or in the past, and 24 never received GH. There was no significant difference in age or body mass index (BMI) (kg/m2) between GH-treated versus non-GH-treated groups. BMI was higher in the deletion group compared to the UPD group (p = 0.05). We identified two individuals who were clinically diagnosed and treated for hypothyroidism, one of whom was on GH supplements. We identified two additional individuals with subclinical hypothyroidism who were not on GH treatment, giving a frequency of 7.6% (4/52) in this cohort of patients. We did not find significant differences in thyroid function (TSH) in the deletion versus UPD groups. We found significant differences in thyroid function, however, between GH-treated and non-GH-treated groups. The mean TSH was lower (2.25 ± 1.17 uIU/M, range 0.03-4.92 uIU/M versus 2.80 ± 1.44 uIU/M, range 0.55-5.33 uIU/M respectively, p = 0.046), and the free T4 levels were significantly higher (1.13 ± 0.70 and 1.03 ± 0.11 ng/dL, respectively, p = 0.05) in the GH-treated individuals compared to non-GH-treated individuals. In this cohort of subjects with PWS, we identified two previously diagnosed individuals with hypothyroidism and two individuals with subclinical hypothyroidism (4/52, 7.6%), three of whom were not receiving GH treatment. We did not find any significant differences in thyroid function between molecular subtypes; however, we found that euthyroid status (lower TSH levels and higher free T4 levels) was significantly higher in individuals who were treated with GH compared to the untreated group. We recommend that individuals with PWS should be screened regularly for thyroid deficiency and start treatment early with GH in view of the potentially lower incidence of thyroid deficiency.
Two siblings suffering from Angelman Syndrome with a novel c.1146T>G mutation in UBE3A: A case report.
Angelman syndrome (AS) is an autosomal dominant neurodevelopmental genetic disease with maternal imprint, which is associated with the presence of the abnormal chromosome 15q11-q13, and the loss of maternal specific expression of ubiquitin-protein ligase E3A (UBE3A). The expression levels of UBE3A depend on the parental origin and exhibit tissue specificity. In normal brain tissues, the maternal UBE3A gene is actively expressed, whereas the paternal UBE3A gene is not. In total, ~85% of pediatric patients with AS present with epilepsy within their 3rd year of life. This condition is usually difficult to control with medical treatment. An 8-year-old female visited the Affiliated Hospital of Jining Medical University due to frequent epilepsy. Her clinical manifestations included specific facial features, moderate mental retardation and frequent seizures. It was interesting to note that her 15-year-old sister exhibited similar clinical manifestations to those of AS. The results of the electroencephalogram and the imaging examinations were also in line with the characteristics of AS. In order to further clarify the diagnosis, all the suspected genes in her sister and in their parents were sequenced. The multiplex ligation-dependent probe amplification project of the Angel/chubby and copy number variation (CNV) sequencing were assessed concomitantly to identify the pathogenic genes responsible for the development of AS. The latter occurs due to the missense mutation c.1146T>G, which results in asparagine replacement by lysine at position 382 (p.Asn382Lys) in exon 7. This amino acid change affects the normal expression of UBE3A; the mutation is a novel mutation, which, to the best of our knowledge, has not been previously reported. Relevant large fragments of mutations and methylation abnormalities were not found in the associated genes. The data further revealed absence of 25-bp repeat mutations at the shear mutation site of exon 1 of the small nuclear ribonucleoprotein polypeptide N gene in the subjects examined. No suspected CNV was found following analysis.
Clinical Utility of Methylation-Specific Multiplex Ligation-Dependent Probe Amplification for the Diagnosis of Prader-Willi Syndrome and Angelman Syndrome.
Prader-Willi syndrome (PWS) and Angelman syndrome (AS) are genomic imprinting disorders that are mainly caused by a deletion on 15q11-q13, the uniparental disomy of chromosome 15, or an imprinting defect. We evaluated the utility of methylation-specific multiplex ligation-dependent probe amplification (MS-MLPA) as a diagnostic tool and for demonstrating the relationship between molecular mechanisms and clinical presentation. We performed MS-MLPA using DNA samples from 93 subjects (45 PWS, 24 AS, and 24 non-PWS/AS controls) who had previously undergone MS-PCR for the diagnosis of PWS/AS. We compared the results of both assays, and patients' clinical phenotypes were reviewed retrospectively. MS-MLPA showed a 100% concordance rate with MS-PCR. Among the 45 PWS patients, 26 (57.8%) had a deletion of 15q11-q13, and the others (42.2%) had uniparental disomy 15 or an imprinting defect. Among the 24 AS patients, 16 (66.7%) had a deletion of 15q11-q13, 7 AS patients (29.2%) had uniparental disomy 15 or an imprinting defect, and one AS patient (4.2%) showed an imprinting center deletion. MS-MLPA has clinical utility for the diagnosis of PWS/AS, and it is superior to MS-PCR in that it can identify the molecular mechanism underlying the disease.
Publicações recentes
Mast cell mediators in hereditary angioedema.
Prenatal Molecular Diagnosis of COL2A1-Associated Stickler Syndrome: Genotype-Phenotype Correlation in a Resource-Limited Healthcare Setting.
Platelet gene signatures detecting pulmonary artery stenosis in patients with pulmonary hypertension.
The global impact of imiglucerase therapy in children with Gaucher disease types 1 and 3: a real-world analysis from the International Collaborative Gaucher Group Gaucher Registry.
Monogenic lupus with SLC7A7 mutations: a retrospective study from a Chinese center.
📚 EuropePMCmostrando 23
Clinical Presentation, Genetics, and Laboratory Testing with Integrated Genetic Analysis of Molecular Mechanisms in Prader-Willi and Angelman Syndromes: A Review.
International journal of molecular sciencesUBE3A reinstatement restores behaviorand proteome in an Angelman syndrome mouse model of imprinting defects.
Molecular autismRelationship of thyroid function with genetic subtypes and treatment with growth hormone in Prader-Willi syndrome.
American journal of medical genetics. Part ATwo siblings suffering from Angelman Syndrome with a novel c.1146T>G mutation in UBE3A: A case report.
Biomedical reportsInvestigation of age-related facial variation among Angelman syndrome patients.
Scientific reportsClinical Utility of Methylation-Specific Multiplex Ligation-Dependent Probe Amplification for the Diagnosis of Prader-Willi Syndrome and Angelman Syndrome.
Annals of laboratory medicine[Prader-Willi and Angelman syndromes: case series diagnosed by MS-MLPA assay].
Revista medica del Instituto Mexicano del Seguro SocialGenotype-Phenotype Correlations in Angelman Syndrome.
GenesPrader-Willi syndrome: reflections on seminal studies and future therapies.
Open biologyCongenital ichthyosis in Prader-Willi syndrome associated with maternal chromosome 15 uniparental disomy: Case report and review of autosomal recessive conditions unmasked by UPD.
American journal of medical genetics. Part AAngelman syndrome genotypes manifest varying degrees of clinical severity and developmental impairment.
Molecular psychiatryA newborn screening pilot study using methylation-sensitive high resolution melting on dried blood spots to detect Prader-Willi and Angelman syndromes.
Scientific reportsCharacterizing spine issues: If offers novel therapeutics to Angelman syndrome.
Developmental neurobiologyAssociation of congenital cardiovascular malformation and neuropsychiatric phenotypes with 15q11.2 (BP1-BP2) deletion in the UK Biobank.
European journal of human genetics : EJHGA placebo-controlled trial of folic acid and betaine in identical twins with Angelman syndrome.
Orphanet journal of rare diseasesGenetic testing for Prader-Willi syndrome and Angelman syndrome in the clinical practice of Guangdong Province, China.
Molecular cytogeneticsA mouse model of Angelman syndrome imprinting defects.
Human molecular genetics[Variation analysis of the number of copies and methylene patterns in region 15q11-q13].
MedicinaAngelman Syndrome due to a Maternally Inherited Intragenic Deletion Encompassing Exons 7 and 8 of the UBE3A Gene.
Cytogenetic and genome researchClinical and genetic aspects of the 15q11.2 BP1-BP2 microdeletion disorder.
Journal of intellectual disability research : JIDRAtypical Angelman syndrome due to a mosaic imprinting defect: Case reports and review of the literature.
American journal of medical genetics. Part AA modified MS-PCR approach to diagnose patients with Prader-Willi and Angelman syndrome.
Molecular biology reportsPrader-Willi syndrome: a review of clinical, genetic, and endocrine findings.
Journal of endocrinological investigationAssociações
Organizações que acompanham esta doença — pra ter apoio e orientação
Ainda não temos associações cadastradas para Síndrome Angelman por anomalia do imprinting no locus 15q11-q13.
É de uma associação que acompanha esta doença? Fale com a gente →
Comunidades
Grupos ativos de quem convive com esta doença aqui no Raras
Ainda não existe comunidade no Raras para Síndrome Angelman por anomalia do imprinting no locus 15q11-q13
Pacientes, familiares e cuidadores se organizam em comunidades pra compartilhar experiências, fazer perguntas e se apoiar. Você pode ser o primeiro.
Tire suas dúvidas
Perguntas, dicas e experiências compartilhadas aqui na página
Participe da discussão
Faça login para postar dúvidas, compartilhar experiências e interagir com especialistas.
Fazer loginDoenças relacionadas
Doenças com sintomas parecidos — ajudam quem ainda está buscando diagnóstico
Referências e fontes
Bases de dados externas citadas neste artigo
Publicações científicas
Artigos indexados no PubMed ligados a esta doença no grafo RarasNet — título, periódico e PMID direto da fonte, sem intermediação de IA.
- Clinical Presentation, Genetics, and Laboratory Testing with Integrated Genetic Analysis of Molecular Mechanisms in Prader-Willi and Angelman Syndromes: A Review.
- UBE3A reinstatement restores behaviorand proteome in an Angelman syndrome mouse model of imprinting defects.
- Relationship of thyroid function with genetic subtypes and treatment with growth hormone in Prader-Willi syndrome.
- Two siblings suffering from Angelman Syndrome with a novel c.1146T>G mutation in UBE3A: A case report.
- Clinical Utility of Methylation-Specific Multiplex Ligation-Dependent Probe Amplification for the Diagnosis of Prader-Willi Syndrome and Angelman Syndrome.
- Mast cell mediators in hereditary angioedema.
- Prenatal Molecular Diagnosis of COL2A1-Associated Stickler Syndrome: Genotype-Phenotype Correlation in a Resource-Limited Healthcare Setting.
- Platelet gene signatures detecting pulmonary artery stenosis in patients with pulmonary hypertension.
- The global impact of imiglucerase therapy in children with Gaucher disease types 1 and 3: a real-world analysis from the International Collaborative Gaucher Group Gaucher Registry.
- Monogenic lupus with SLC7A7 mutations: a retrospective study from a Chinese center.
Bases de dados e fontes oficiais
Identificadores e referências canônicas usadas para montar este verbete.
- ORPHA:411515(Orphanet)
- MONDO:0018462(MONDO)
- GARD:21733(GARD (NIH))
- Variantes catalogadas(ClinVar)
- Busca completa no PubMed(PubMed)
- Q55788101(Wikidata)
Dados compilados pelo RarasNet a partir de fontes abertas (Orphanet, OMIM, MONDO, PubMed/EuropePMC, ClinicalTrials.gov, DATASUS, PCDT/MS). Este conteúdo é informativo e não substitui avaliação médica.
Conteúdo mantido por Agente Raras · Médicos e pesquisadores podem colaborar